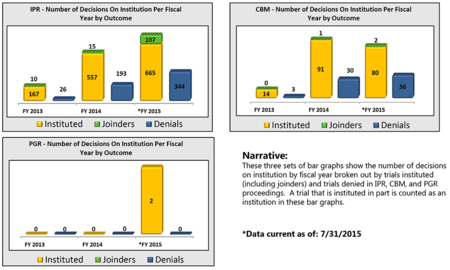
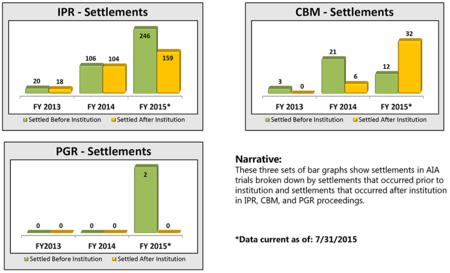
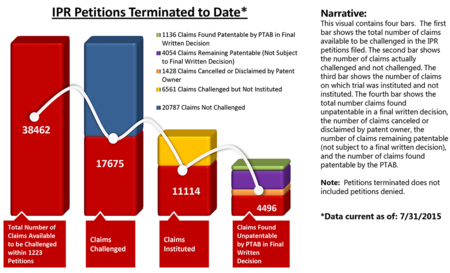
By Michael Borella —
 On July 30, 2015, the U.S. Patent and Trademark Office updated its subject matter eligibility guidance ("Eligibility Update"). This update provides recommendations and resources for examiners in addition to those in the Office's 2014 Interim Guidance on Subject Matter Eligibility. In particular, the Office addressed several broad themes from comments it received on the Interim Guidance. The focus of this article is on how the Eligibility Update potentially impacts claims that may incorporate abstract ideas.
On July 30, 2015, the U.S. Patent and Trademark Office updated its subject matter eligibility guidance ("Eligibility Update"). This update provides recommendations and resources for examiners in addition to those in the Office's 2014 Interim Guidance on Subject Matter Eligibility. In particular, the Office addressed several broad themes from comments it received on the Interim Guidance. The focus of this article is on how the Eligibility Update potentially impacts claims that may incorporate abstract ideas.
In the Eligibility Update, the Office provides supplementary analyses of hypothetical claims, some patent eligible and some not. However, unlike the abstract idea examples that the Office published in January of this year, these analyses include sets of claims, for a given invention, that were both eligible and ineligible. Such examples may prove to be quite valuable, as they allow applicants to focus in on features and claim language that can transform a claim that is too abstract into one that meets the subject matter requirements.
The new example claims include some directed to distribution of stock quotes, graphical user interfaces, and remote loading of Basic Input / Output System (BIOS) bootstrap code on a computer, along other hypotheticals. Unsurprisingly, the ineligible claims tend to be pure business method or disembodied processes, while the eligible claims involve a more substantial change in the operation of a computer.
When discussing these examples, the Office notes that the second prong of the patent-eligibility test from Alice Corp. v. CLS Bank Int'l (for a claim incorporating an abstract idea, determining whether the claim recites "significantly more than a patent upon the ineligible concept itself") requires that examiners consider the additional claim elements both individually and as an ordered combination. The Office writes that "even if an element does not amount to significantly more on its own . . . it can still amount to significantly more when considered in combination with the other elements of the claim." Particularly, the Office states that, in some cases, "generic computer components that individually perform merely generic computer functions . . . are able in combination to perform functions that are not generic computer functions and that amount to significantly more." This is welcome news, as examiners all too often view the elements of claims in isolation when evaluating patent eligibility (as well as other patentability requirements for that matter).
The Office also provides supplementary material on how examiners should endeavor to identify abstract ideas. Due to the Supreme Court's decision in Alice to purposely not define abstract ideas other than by example, the Office is now following the strategy employed by some Federal Circuit and district court judges — compare the claims at issue to those found in relevant case law. This "walks like a duck" test expands the two-prong Alice inquiry into a more complex animal. The examiner reviews the claims in light of the several dozen Supreme Court and Federal Circuit decisions finding claims ineligible under 35 U.S.C. § 101 (e.g., Alice, Bilski v. Kappos, Ultramercial, Inc. v. Hulu, LLC, etc.), as well as the two decisions finding challenged claims eligible (Diamond v. Diehr and DDR Holdings, LLC v. Hotels.com, L.P.). While there are several other examples of claims that pass muster under § 101 in this Eligibility Update, Diehr and DDR Holdings are the only two cases addressing eligible claims involving abstract ideas that have either been blessed by the Supreme Court or decided post-Alice by the Federal Circuit. Once the examiner has conducted the comparison, an eligibility decision is made based on the likeness (or lack thereof) of the examined claims to the relevant case law.
This process could be faulted as being more than a little inexact. Ultimately, however, the finger must be pointed back to the Supreme Court. The Office is merely following the example of the Federal Circuit, the panels thereof struggling to interpret Alice and its predecessors.
But the Eligibility Update does provide some clarity with respect to the intersection of § 101 with §§ 102 and 103. The Office writes "[w]hen identifying abstract ideas, examiners should keep in mind that judicial exceptions need not be old or long‐prevalent, and that even newly discovered judicial exceptions are still exceptions, despite their novelty." Pointing to Parker v. Flook, Mayo Collaborative Svcs. v. Prometheus Labs, and Ultramercial as examples, the Office contends that the claims of these cases tied up "basic tools of scientific and technological work that lie beyond the domain of patent protection." Thus, an argument rebutting a § 101 rejection or invalidity contention by pointing out the novelty of the claimed invention is likely dead on arrival.
The Office then goes on to discuss four particular categories of claims that are likely to be viewed as abstract. The Office made it clear that these categories are not mutually exclusive, and that claims can belong to two or more.
• Fundamental economic practices, in the Office's view, are "concepts relating to the economy and commerce, such as agreements between people in the form of contracts, legal obligations, and business relations." But, "[t]he term 'fundamental' is used in the sense of being foundational or basic, and not in the sense of necessarily being 'old' or 'well‐known.'" The Supreme Court, however, made a distinction between claims directed to "fundamental economic practices" and "fundamental truths" in Alice. The former is "long prevalent in our system of commerce" (i.e., in the prior art), while the latter "an original cause; a motive" that "exist[s] in principle apart from any human action." The Court differentiated between these two, noting that the claims of Bilski involved "a method of organizing human activity, not a truth about the natural world that has always existed."
• Regarding the organization of human activity, the Office concedes that "not all methods of organizing human activity are abstract ideas." For instance, human operation of machines does not fall into this category. Instead, these methods may include "concepts relating to interpersonal and intrapersonal activities, such as managing relationships or transactions between people, social activities, and human behavior; satisfying or avoiding a legal obligation; advertising, marketing, and sales activities or behaviors; and managing human mental activity."
• An idea of itself, according to the Office, is "an idea standing alone such as an uninstantiated concept, plan or scheme, as well as a mental process (thinking) that can be performed in the human mind, or by a human using a pen and paper." Examples of such ideas include comparisons of data, performing tests and thinking about the results, using categories to process information, and the display of advertising in exchange for access to media of Ultramercial.
• Mathematical relationships and formulas, as another example, are "used to describe mathematical concepts such as mathematical algorithms, mathematical relationships, mathematical formulas, and calculations." Instances of claims addressing such relationships or formulas can be found in the Supreme Court cases of Benson v. Gottschalk, Flook, Diehr, and Bilski.
The Office adds some further clarity when it discusses the requirements of a prima facie case of patent ineligibility. Many § 101 rejections coming out of the Office are conclusory in nature, leaving the applicant with little substance to rebut. This has led commenters to request that the Office clarify the examiner's burden in this regard.
The Office begins by stating that "performing a thorough analysis and writing a clear rejection is a critical part of satisfying the examiner's burden." Moreover, "the examiner's burden is met by clearly articulating the reason(s) why the claimed invention is not eligible, for example by providing a reasoned rationale that identifies the judicial exception recited in the claim and why it is considered an exception, and that identifies the additional elements in the claim (if any) and explains why they do not amount to significantly more than the exception." In doing so, the examiner "may rely, where appropriate, on the knowledge generally available to those in the art, on the case law precedent, on applicant's own disclosure, or on evidence."
Addressing whether subject matter eligibility decisions require factual findings, the Office appears to answer this question in the negative. Particularly, "determination of whether a claim is eligible [is] a question of law" and "courts do not rely on evidence that a claimed concept is a judicial exception, and in most cases resolve the ultimate legal conclusion on eligibility without making any factual findings." Instead, the Office instructs examiners to "rely on what the courts have recognized, or those in the art would recognize, as elements that are well‐understood, routine and conventional" — in other words, to apply the "walks like a duck" test.
Notably, with reference to the second prong of the Alice framework, the Office indicates that evidence is not necessarily "required to support a finding that the additional elements were well‐understood, routine or conventional, but [examiners should] treat the issue as a matter appropriate for judicial notice." Thus, "a rejection should only be made if an examiner relying on his or her expertise in the art can readily conclude [by applying second prong of the Alice] that the additional elements do not amount to significantly more."
One of the more mysterious aspects of § 101 post-Alice is the role of preemption in the analysis. The Supreme Court stated in that case that the "preemption concern . . . undergirds our §101 jurisprudence." The Office, however, believes that "questions of preemption are inherent in the two‐part framework from Alice Corp. and Mayo . . . , and are resolved by using this framework to distinguish between preemptive claims" and those that are not. But, as set forth in Ultramercial, "absence of complete preemption does not guarantee that a claim is eligible." Here, the Office does not resolve the preemption issue, but instead chooses not to address it directly at this point.
Finally, the Office tackles concerns of when its streamlined § 101 analysis may be used. The Interim Guidance allows examiners to skip the Alice test for "claims that clearly do not seek to tie up any judicial exception such that others cannot practice it." The Office reiterates that the outcome of the patent-eligibility analysis will be the same whether the streamlined analysis or the full Alice procedure is used. Notably, examiners do not need to indicate when the streamlined analysis has been employed, as "the record may reflect the conclusion of eligibility simply by the absence of an eligibility rejection or may include clarifying remarks."
 The Federal Circuit's Ariosa v. Sequenom decision handed down earlier this summer marked the apex (or nadir, according to your prejudices) of the current trend to limit the scope of patent eligible subject matter. On Thursday, twelve amicus curiae briefs were filed in support of Sequenom's petition to the full court for rehearing en banc. We will post on the substance of these briefs in later posts; for now the posts can be accessed here:
The Federal Circuit's Ariosa v. Sequenom decision handed down earlier this summer marked the apex (or nadir, according to your prejudices) of the current trend to limit the scope of patent eligible subject matter. On Thursday, twelve amicus curiae briefs were filed in support of Sequenom's petition to the full court for rehearing en banc. We will post on the substance of these briefs in later posts; for now the posts can be accessed here: