By Donald Zuhn —
 Earlier today, the U.S. Patent and Trademark Office published its interim guidance regarding the examination of claims for subject matter eligibility under 35 U.S.C. § 101 (which the Office announced yesterday) in the Federal Register (79 Fed. Reg. 74618). The Federal Register notice, entitled "2014 Interim Guidance on Patent Subject Matter Eligibility," outlines an examination process that supplements the Office's "Preliminary Examination Instructions in view of the Supreme Court Decision in Alice Corporation Pty. Ltd. v. CLS Bank International," which was issued in June (see "USPTO Issues Preliminary Examination Instructions Regarding Alice Corp. v. CLS Bank International"), and supersedes the Office's "Procedure for Subject Matter Eligibility Analysis of Claims Reciting or Involving Laws of Nature/Natural Principles, Natural Phenomena, and/or Natural Products, which was issued in March (see "USPTO Issues Guidance for Analyzing Subject Matter Eligibility of Claims Reciting Laws of Nature/Natural Principles, Natural Phenomena or Natural Products") ("Myriad/Mayo guidance"). The notice indicates that the interim guidance takes effect today, and includes a request for public comment.
Earlier today, the U.S. Patent and Trademark Office published its interim guidance regarding the examination of claims for subject matter eligibility under 35 U.S.C. § 101 (which the Office announced yesterday) in the Federal Register (79 Fed. Reg. 74618). The Federal Register notice, entitled "2014 Interim Guidance on Patent Subject Matter Eligibility," outlines an examination process that supplements the Office's "Preliminary Examination Instructions in view of the Supreme Court Decision in Alice Corporation Pty. Ltd. v. CLS Bank International," which was issued in June (see "USPTO Issues Preliminary Examination Instructions Regarding Alice Corp. v. CLS Bank International"), and supersedes the Office's "Procedure for Subject Matter Eligibility Analysis of Claims Reciting or Involving Laws of Nature/Natural Principles, Natural Phenomena, and/or Natural Products, which was issued in March (see "USPTO Issues Guidance for Analyzing Subject Matter Eligibility of Claims Reciting Laws of Nature/Natural Principles, Natural Phenomena or Natural Products") ("Myriad/Mayo guidance"). The notice indicates that the interim guidance takes effect today, and includes a request for public comment.
With respect to the Office's Myriad/Mayo guidance, the Office's notice states (in a footnote) that the analysis outlined in the interim guidance:
[D]iffers from the [analysis outlined in the Myriad/Mayo guidance] in certain respects. Note, for example, the test for determining whether a claim is directed to a "product of nature" exception is separated from the analysis of whether the claim includes significantly more than the exception. Also, the application of the overall analysis is based on claims directed to judicial exceptions (defined as claims reciting the exception, i.e., set forth or described), rather than claims merely "involving" an exception. For instance, process claims that merely use a nature-based product are not necessarily subject to an analysis for markedly different characteristics. Additionally, the markedly different analysis focuses on characteristics that can include a product's structure, function, and/or other properties as compared to its naturally occurring counterpart in its natural state. [Emphasis in original.]
The differences between the interim guidance and Myriad/Mayo guidance correspond to statements made by Office representatives over the past few months (see "Docs @ BIO: USPTO Provides Update on Myriad-Mayo Guidance"; "USPTO Expected to Issue Revised Myriad-Mayo Guidance in October"; "USPTO Outlines Changes to Myriad-Mayo Guidance at BIO Symposium"; and "USPTO Provides Update on Status of Revised Myriad-Mayo Guidance").
As for the Office's preliminary Alice Corp. guidance, the notice points out that the preliminary Alice Corp. guidance superseded some subject matter eligibility guidance in the MPEP (§§ 2106(II)(A) and 2106(II)(B)) and that the interim guidance supersedes some subject matter eligibility guidance in the MPEP (§ 2105, in part, and § 2106.01), but that "[e]xaminers should continue to follow the MPEP for all other examination instructions."
The notice indicates that the interim guidance "offers a comprehensive view of subject matter eligibility in line with Alice Corp, Myriad, Mayo, and the related body of case law," and is responsive to the public feedback the Office has received concerning the Myriad/Mayo guidance and preliminary Alice Corp. guidance. While the Office acknowledges that "there may be variations in the precise contours of the analysis for subject matter eligibility that will still achieve the same end result," the Office asserts that the analysis outlined in the interim guidance will "promote[] examination efficiency and consistency across all technologies."
Following the presentation of a flowchart, which sets forth the eligibility test for both products and processes, the interim guidance is divided into five parts: (1) a two-part analysis for determining whether a claim is directed to a judicial exceptions, (2) a brief reminder about complete examination, (3) examples of claims, based on Supreme Court decisions, analzyed using the interim guidance analysis, and (4) summaries of Supreme Court and Federal Circuit decisions relating to laws of nature, natural phenomena, and abstract ideas. Today, we will provide an overview of the interim guidance and a discussion of the first two parts of the guidance. More detailed analyses of Parts III and IV of the interim guidance, as well as additional perspectives of how the interim guidance will impact applicants and practitioners in both the Myriad/Mayo space and Alice space, will be provided in subsequent posts.
The analysis to be used in determining "whether a claim is drawn to patent-eligible subject matter" is illustrated in the interim guidance by the following flowchart (click on flowchart to expand):
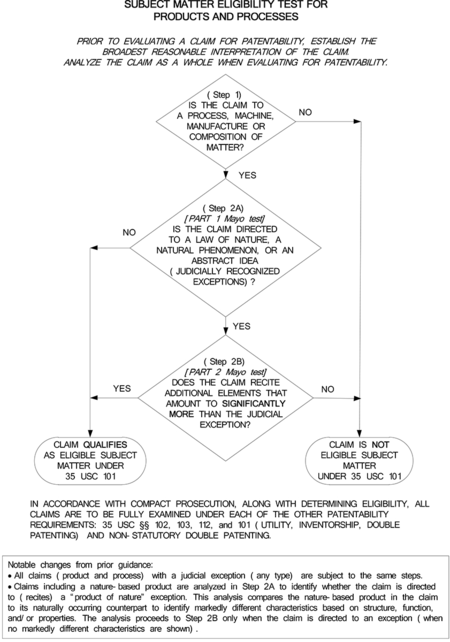
According to the interim guidance, the current two-step analysis for determining subject matter eligibility under 35 U.S.C. § 101, as set forth in MPEP § 2106, consists of: (1) determining whether the claimed invention is "directed to one of the four statutory categories" (i.e., process, machine, manufacture, or composition of matter), and (2) determining whether the claimed invention is "wholly directed to subject matter encompassing a judicially recognized exception." In the flowchart included in the interim guidance, the first step of the above analysis is shown in the first diamond (labeled "Step 1"), and the second step of the analysis is shown in the second and third diamonds (labeled "Step 2A" and "Step 2B"). The interim guidance notes that the second step has been divided into two steps (or diamonds) in the flowchart to correspond with the two-step (or two-part) analysis from Alice Corp. (which was set forth earlier in Mayo). These last two steps/diamonds are the subject of the interim guidance.
The interim guidance indicates that in Step 2A (the second diamond on the flowchart), which corresponds to part 1 of the Alice Corp./Mayo test, the examiner shall "[d]etermine whether the claim is directed to a law of nature, a natural phenomenon, or an abstract idea (judicial exceptions)." As for Step 2B (the third diamond on the flowchart), which corresponds to part 2 of the Alice Corp./Mayo test, the interim guidance indicates that the examiner shall "[d]etermine whether any element, or combination of elements, in the claim is sufficient to ensure that the claim amounts to significantly more than the judicial exception."
With regard to Step 2A, the interim guidance states that if the examiner determines that the claimed invention is not directed to any judicial exceptions, the claim is eligible and needs no further eligibility analysis. In discussing this step, the interim guidance states that "[a] claim is directed to a judicial exception when a law of nature, a natural phenomenon, or an abstract idea is recited (i.e., set forth or described) in the claim."
As for the judicial exceptions, the interim guidance indicates that "[l]aws of nature and natural phenomena, as identified by the courts, include naturally occurring principles/substances and substances that do not have markedly different characteristics compared to what occurs in nature." The notice then lists several types of concepts that courts have found to be laws of nature and natural phenomena: an isolated DNA (Association for Molecular Pathology v. Myriad Genetics, Inc.), a correlation that is the consequence of how a certain compound is metabolized by the body (Mayo Collaborative Services v. Prometheus Laboratories, Inc.), electromagnetism to transmit signals (O'Reilly v. Morse), and the chemical principle underlying the union between fatty elements and water (Tilghman v. Proctor).
With respect to abstract ideas, the interim guidance indicates that "[a]bstract ideas have been identified by the courts by way of example, including fundamental economic practices, certain methods of organizing human activities, an idea 'of itself,' and mathematical relationships/formulas." The notice then lists several types of concepts that courts have found to be abstract ideas: mitigating settlement risk (Alice Corp. v. CLS Bank); hedging (Bilski v. Kappos); creating a contractual relationship (buySAFE, Inc. v. Google, Inc.); using advertising as an exchange or currency (Ultramercial, LLC v. Hulu, LLC and WildTangent); processing information through a clearinghouse (Dealertrack Inc. v. Huber); comparing new and stored information and using rules to identify options (SmartGene, Inc. v. Advanced Biological Laboratories, SA); using categories to organize, store and transmit information (Cyberfone Systems v. CNN Interactive Group); organizing information through mathematical correlations (Digitech Image Tech., LLC v. Electronics for Imaging, Inc.); managing a game of bingo (Planet Bingo, LLC v. VKGS LLC); the Arrhenius equation for calculating the cure time of rubber (Diamond v. Diehr); a formula for updating alarm limits (Parker v. Flook); a mathematical formula relating to standing wave phenomena (Mackay Radio & Telegraph Co. v. Radio Corp. of America); and a mathematical procedure for converting one form of numerical representation to another (Gottschalk v. Benson).
In applying the Step 2A analysis to nature-based products, the interim guidance notes that an examiner should determine whether a nature-based product limitation in a claim needs to be evaluated using a markedly different characteristic analysis, and if so, to carry out that analysis. (In a footnote, the notice recognizes that "[t]his revised analysis represents a change from prior guidance, because now changes in functional characteristics and other non-structural properties can evidence markedly different characteristics, whereas in the [Myriad/Mayo guidance] only structural changes were sufficient to show a marked difference.")
The notice explains that "[a] nature-based product can be claimed by itself (e.g., 'a Lactobacillus bacterium') or as one or more limitations of a claim (e.g., 'a probiotic composition comprising a mixture of Lactobacillus and milk in a container')," and indicates that in the latter case, "the markedly different characteristics analysis should be applied to the resultant nature-based combination, rather than its component parts." According to the notice, the markedly different characteristics analysis compares the nature-based product limitation to its naturally occurring counterpart in its natural state, or to the closest naturally occurring counterpart when there is no naturally occurring counterpart to the nature-based product. The interim guidance states that "[m]arkedly different characteristics can be expressed as the product's structure, function, and/or other properties," noting that "even a small change can result in markedly different characteristics from the product's naturally occurring counterpart." The interim guidance also states that:
In accordance with this analysis, a product that is purified or isolated, for example, will be eligible when there is a resultant change in characteristics sufficient to show a marked difference from the product's naturally occurring counterpart. If the claim recites a nature-based product limitation that does not exhibit markedly different characteristics, the claim is directed to a "product of nature" exception (a law of nature or naturally occurring phenomenon), and the claim will require further analysis to determine eligibility based on whether additional elements add significantly more to the exception.
The notice provides several examples of the types of characteristics considered by the courts when determining whether there is a marked difference, including: biological or pharmacological functions or activities (citing Funk Bros. Seed Co. v. Kalo Inoculant Co., Diamond v. Chakrabarty, In re King, and Myriad); chemical and physical properties (citing Parke-Davis & Co. v. H.K. Mulford Co. and Funk Bros.; phenotype, including functional and structural characteristics (citing In re Roslin Inst.); and structure and form, whether chemical, genetic or physical (citing Chakrabarty, Parke-Davis, and Myriad).
The interim guidance notes that "[i]f the claim includes a nature-based product that has markedly different characteristics, the claim does not recite a 'product of nature' exception and is eligible . . . unless the claim recites another exception (such as a law of nature or abstract idea, or a different natural phenomenon)." Otherwise, the examiner must then proceed to Step 2B of the analysis.
In Step 2B, the examiner must "determine whether the elements of the claim, considered both individually and as an ordered combination, are sufficient to ensure that the claim as a whole amounts to significantly more than the exception itself," which the interim guidance characterizes as "a search for an 'inventive concept.'" The notice cautions that "[i]t is important to consider the claim as whole," as "[i]ndividual elements viewed on their own may not appear to add significantly more to the claim, but when combined may amount to significantly more than the exception." The notice lists examples of considerations identified by the Supreme Court for determining whether a claim with additional elements amounts to significantly more than the judicial exception itself, including limitations that may be enough to qualify as "significantly more" when recited in a claim with a judicial exception and limitations that were found not to be enough to qualify as "significantly more" (see page 74624 of notice).
If after the Step 2B analysis, the claim is found to be directed to patent-ineligible subject matter, the interim guidance indicates that the examiner should "identify the exception by referring to where it is recited (i.e., set forth or described) in the claim and explain why it is considered an exception," and "[t]hen, if the claim includes additional elements, identify the elements in the rejection and explain why they do not add significantly more to the exception."
Part I of the interim guidance concludes with a brief discussion of claims reciting a plurality of exceptions and a streamlined eligibility analysis. With respect to the former concept, the notice states that for claims directed to a plurality of exceptions, examiners should conduct the eligibility analysis for one of the exceptions. The interim guidance suggests that "if the claim fails under Step 2B for one exception, the claim is ineligible, and no further eligibility analysis is needed." As for streamlined analysis, the interim guidance explains that this analysis "can be used for a claim that may or may not recite a judicial exception but, when viewed as a whole, clearly does not seek to tie up any judicial exception such that others cannot practice it." The notice includes four examples of subject matter for which the streamlined analysis could be used: (1) a robotic arm assembly having a control system that operates using certain mathematical relationships (which the notice states is clearly not an attempt to tie up use of the mathematical relationships), (2) an artificial hip prosthesis coated with a naturally occurring mineral (which the notice states is not an attempt to tie up the mineral), (3) a cellphone with an electrical contact made of gold, (4) a plastic chair with wood trim.
Part II of the interim guidance reminds examiners that "[r]egardless of whether a rejection under 35 U.S.C. 101 is made, a complete examination should be made for every claim under each of the other patentability requirements: 35 U.S.C. 102, 103, 112, and 101 (utility, inventorship and double patenting) and nonstatutory double patenting."
Part III of the interim guidance provides examples, based on Supreme Court decisions, where the examination procedure set forth in the interim guidance is used to analyze claims for subject matter eligibility. The six examples include:
Example 1. Diamond v. Chakrabarty (U.S. Patent No. 4,259,444)
Example 2. Association for Molecular Pathology v. Myriad Genetics, Inc. (U.S. Patent No. 5,747,282)
Example 3. Diamond v. Diehr (U.S. Patent No. 4,344,142)
Example 4. Parker v. Flook
Example 5. Mayo v. Prometheus (U.S. Patent No. 6,355,623)
Example 6. Alice Corp. v. CLS Bank (U.S. Patent Nos. 5,970,479 and 7,725,375)
As noted above, Patent Docs will provide more detailed analyses of Part III in subsequent posts (readers may note that detailed analyses of several of the cases used in Part III have been previously provided in this space, including Myriad (here and here), Mayo, and Alice Corp. (here and here)).
Part IV of the interim guidance provides brief summaries of Supreme Court and Federal Circuit decisions in which the claims at issue were analyzed with respect to judicial exceptions for subject matter eligibility. This section of the interim guidance provides summaries of the following Supreme Court decisions:
• O'Reilly v. Morse (U.S. Reissue Patent No. RE 117)
• Tilghman v. Proctor (U.S. Patent No. 11,766)
• Mackay Radio & Telegraph Co. v. Radio Corp. of America (U.S. Patent No. 1,974,387)
• Gottschalk v. Benson
• Bilski v. Kappos
This section also provides summaries of the following Federal Circuit decisions (divided into two groups: abstract idea decisions issued prior to the Supreme Court's Alice Corp. decision and abstract idea decisions issued since the Alice Corp. decision):
• SiRF Technology v. ITC (U.S. Patent No. 6,417,801)
• Research Corp. Tech. v. Microsoft Corp. (U.S. Patent No. 5,111,310)
• Dealertrack Inc. v. Huber (U.S. Patent No. 7,181,427)
• SmartGene, Inc. v. Advanced Biological Laboratories, SA (U.S. Patent No. 6,081,786)
• Cyberfone Systems v. CNN Interactive Group (U.S. Patent No. 8,019,060)
• Digitech Image Tech., LLC v. Electronics for Imaging, Inc. (U.S. Patent No. 6,128,415)
• Planet Bingo, LLC v. VKGS LLC (U.S. Patent No. 6,398,646)
• buySAFE, Inc. v. Google, Inc. (U.S. Patent No. 7,644,019)
• Ultramercial, LLC v. Hulu, LLC and WildTangent (U.S. Patent No. 7,346,545)
• DDR Holdings, LLC v. Hotels.com, L.P. (U.S. Patent No. 7,818,399)
As noted above, Patent Docs will provide more detailed analyses of Part IV in subsequent posts (readers may note that detailed analyses of many of the cases discussed in Part IV have been previously provided in this space, including the following cases: Bilksi, SmartGene, Cyberfone Systems, Digitech Image Tech., Planet Bingo, Ultramercial, and DDR Holdings).
The notice also indicates that explanatory training examples relating to nature-based products, replacing the examples in the Myriad/Mayo guidance, will be released (those examples were posted on the Office's interim guidance website on Monday; Patent Docs will provide a more detailed analysis of the nature-based products examples in a subsequent post). The notice further indicates that explanatory example sets relating to claims that do and do not amount to significantly more than a judicial exception are being developed and will be issued at a future date (Drew Hirshfeld, U.S. Patent and Trademark Office Deputy Commissioner for Patent Examination Policy, indicated during a conference call on Monday that those examples were expected to be released in one to two weeks).
In addition to outlining the Office's interim guidance on subject matter eligibility, the notice includes a request for public comment regarding the interim guidance as well as suggestions for examples to be used in examiner training and in the explanatory example sets that have been (and will be) posted on the Office's interim guidance website. Echoing comments made by Mr. Hirshfeld during Monday's conference call, the notice states that "[i]mplementation of examination guidance on eligibility will be an iterative process continuing with periodic supplements based on developments in patent subject matter eligibility jurisprudence and public feedback." Written comments regarding the interim guidance and suggesting additional examples can be sent by e-mail to: 2014_interim_guidance@uspto.gov. The deadline for submitting written comments is March 16, 2015.
The notice also indicates that the Office will be holding a public forum in mid-January 2015 to discuss the interim guidance and receive additional oral input on the interim guidance. Mr. Hirshfeld indicated during Monday's conference call that the forum would take place on January 21, 2015. The Office's interim guidance website further indicates that the eligibility forum will take place from 1:00 to 5:00 pm (EST) on that date in the USPTO's Madison Auditorium North (Concourse Level) in Alexandria, VA. Information regarding the forum, which will be accessible via WebEx, can be found on the interim guidance website.

Leave a comment