By Kevin E. Noonan –
 Not surprisingly, the Federal Circuit visited upon Plaintiff/Appellant PureCircle two of the Four Horsemen of the Biotech Patent Apocalypse* in a decision affirming the District Court's invalidation of the claims asserted against Defendant SweeGen in PureCircle USA Inc. v. SweeGen, Inc.
Not surprisingly, the Federal Circuit visited upon Plaintiff/Appellant PureCircle two of the Four Horsemen of the Biotech Patent Apocalypse* in a decision affirming the District Court's invalidation of the claims asserted against Defendant SweeGen in PureCircle USA Inc. v. SweeGen, Inc.
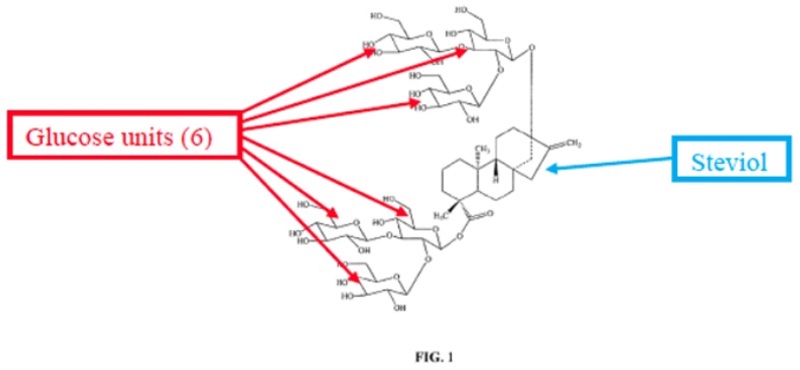
To recap, PureCircle sued SweeGen for infringing U.S. Patent Nos. 9,243,273 (claims 1-14) and 10,485,257 (claims 1-7) over methods for making particular glucosylated forms of the natural sweetener steviol obtained from the Stevia rebaudiana plant. The plant produces a variety of rebaudioside variants having various levels of glycosylation; the most predominant of these in the plant is termed "RebA" but this is not the most commercially valuable form. That form, termed variably "RebX" and "RebM" contains six glucose residues on the steviol core, illustrated in the opinion by this diagram:
Claim 1 of each of the asserted patents are relevant to the decision of both the District Court and the Federal Circuit:
Claim 1 of the '257 patent:
A method for adding at least one glucose unit to a steviol glycoside substrate to provide a target steviol glycoside, comprising contacting the steviol glycoside substrate with a recombinant biocatalyst protein enzyme comprising UDP-glucosyltransferase, wherein the target steviol glycoside is Rebaudioside X.
Claim 1 of the '273 patent:
A method for making Rebaudioside X comprising a step of converting Rebaudioside D to Rebaudioside X using a UDP-glucosyltransferase, wherein the conversion of Rebaudioside D to Rebaudioside X is at least about 50% complete.
The District Court granted summary judgment against PureCircle, finding that claims 1-5 of the '257 patent and claims 1-11 and 14 of the '273 patent were invalid for reciting patent-ineligible subject matter under 35 U.S.C. § 101. According to the Court, the claims were directed to the natural law of converting rebaudiosides, and specifically RebD, into RebM. In the Court's view, "there is no dispute that the conversion of steviol glycosides and Reb D to Reb M using UGT enzymes is a natural process." The Court found this characterization to be supported by disclosure in the specification, which was expressed as disclosing a "biocatalytic process," defined as "the use of natural catalysts, such as protein enzymes, to perform chemical transformations on organic compounds" (emphasis added). The Court relied on Athena Diagnostics, Inc. v. Mayo Collaborative Services, LLC (Fed. Cir. 2019), for the proposition that "[s]ynthetically-created chemical compositions that are structurally and functionally identical to their naturally-occurring counterparts . . . are not patent eligible" and the admission in the specification that the recombinant enzymes were structurally and functionally "identical" to the naturally occurring enzymes. The District Court rejected the distinction PureCircle attempted to draw for "method of preparation" claims analogous to CellzDirect and expressly with regard to any suggestion that such claims are per se patent eligible, as being inconsistent with precedent particularly the Supreme Court's rationale in Alice v CLS Bank (2014).
The District Court also found that all asserted claims were invalid for failure to satisfy the written description requirement of 35 U.S.C. § 112(a), based on the parties' stipulation that the UGT enzyme recited for converting steviol glycosides and Reb D to Reb M was defined as "[a] type of enzyme that is capable of transferring a glucose unit from a uridine diphosphate glucose molecule to a steviol glycoside molecule." From this, the Court held that the enzyme was functionally (as opposed to structurally) defined and thus invalid under Juno Therapeutics, Inc. v. Kite Pharma, Inc., 10 F.4th 1330, 1337 (Fed. Cir. 2021). Pure Circle appealed.
The Federal Circuit affirmed, in an opinion by Judge Dyk joined by Judges Schall and Stark. The opinion first addressed the Section 112(a) grounds for invalidating all claims of the patents, reciting the requirement that, for a genus claim the required written description must disclose "either a representative number of species falling within the scope of the genus or structural features common to the members of the genus so that one of skill in the art can 'visualize or recognize' the members of the genus," the standard first set forth by Judge Lourie in Regents of the Univ. of California v. Eli Lilly & Co., 119 F.3d 1559, 1568–69 (Fed. Cir. 1997). The panel agreed with the District Court's characterization of the '257 and '273 patent claims as being "genus claims using functional language." SweeGen argued (echoing Sanofi's similar arguments for the enablement requirement in Amgen v. Sanofi) that the number of UGT enzymes based on the one expressly disclosed enzyme (UGT76G1) that would satisfy the functional definition was "at least one trillion" (using calculations recited in the panel's opinion), noting that there were 733 UGT enzymes known in 2012 from which various permutations of mutations could produce the cited astronomical number. Accordingly, SweeGen argued, the common '257 and '273 patent specification did not disclose a representative number of species nor did they disclose any common structural feature(s). Although PureCircle was able to argue that, rather than trillions of species there might be as many as 9,000 this argument was unpersuasive to both the District Court and the Federal Circuit; the Court opined that characterization of 9,000 species comprised "extensive trial and error testing" and the need thereof was contrary to finding that an adequate written description was provided in the specification, citing Novozymes A/S v. DuPont Nutrition Biosciences APS, 723 F.3d 1336, 1346 (Fed. Cir. 2013). In addition, the Court was unconvinced by PureCircle's argument that the one disclosed UGT enzyme was sufficient to satisfy the written description requirement because it disclosed (purportedly inherently) the active site capable of catalyzing the conversion reaction(s). The possibility that there may be other (unknown) enzymes capable of catalyzing the conversion reaction(s) and the absence of any express disclosure of the structure of UGT76G1 convinced the Federal Circuit that the situation here was analogous to the broad disclosure of scFv molecules in Juno v. Kite and thus that the written description requirement based on structural features was not satisfied. And the opinion notes that as in Ariad v. Eli Lilly & Co.:
Such claims merely recite a description of the problem to be solved while claiming all solutions to it and . . . cover any compound later actually invented and determined to fall within the claim's functional boundaries—leaving it to the pharmaceutical industry to complete an unfinished invention.
The opinion also cites more recent cases, including AbbVie Deutschland GmbH & Co., KG v. Janssen Biotech, Inc., 759 F.3d 1285, 1300 (Fed. Cir. 2014), and Idenix Pharms. LLC v. Gilead Scis. Inc., 941 F.3d 1149, 1164 (Fed. Cir. 2019), in support of this conclusion.
The convergence of the District Court's invalidity determinations based on Sections 101 and 112(a) arose in the opinion with regard to claim 14 of the '273 patent, which is limited to the expressly disclosed UGT76G1 species of UGT enzyme. The Federal Circuit opinion turns in this regard to the Section 101 analysis, affirming the District Court's finding of invalidity for claim 14. As set forth in the opinion, the Court held that "[t]o the extent that claim 14 claims a 'method for making Rebaudioside X comprising a step of converting Rebaudioside D to Rebaudioside X using a UDPglucosyltransferase' it claims a natural phenomenon," thus satisfying the first step of the Mayo/Alice formula for finding Section 101 invalidity. In response to PureCircle's argument that the patentable distinction in this claim is that when in purified form the enzyme can achieve conversion of at least 50% of RebD to RebM (which is not achieved in the natural state), the opinion asserts that "[t]he problem with PureCircle's argument is that the 50% completion is itself an abstract idea." Citing SAP Am., Inc. v. InvestPic, LLC, 898 F.3d 1161, 1167 (Fed Cir. 2018), the Federal Circuit asserts that "claim 14 of the '273 patent 'd[id] not specify how to achieve a particular purity or conversion percentage; rather, [it] only recite[s] the resulting percentages,'" quoting the District Court's opinion. This portion of the opinion illustrates nicely the rhetorical gymnastics that the entire edifice of subject matter eligibility jurisprudence has created; after all, taken at face value this analysis introduces a requirement that the mechanistic details of how a chemical reaction catalyzed by an enzyme be disclosed, rather than merely demonstrating that the enzyme in the presence of starting compounds can produce the desired product. Not surprisingly in view of this heretofore unknown requirement, the opinion notes that "PureCircle made no step two Alice/Mayo arguments before us or the district court" and thus affirmed the District Court's finding of invalidity for claim 14.
The history of the Federal Circuit's explication of the Supreme Court's subject matter eligibility opinions is replete with decisions and rationales that are not for the faint-hearted, wherein methods for producing truck axles, Am. Axle & Mfg., Inc. v. Neapco Holdings LLC, 967 F.3d 1285, 1292 (Fed. Cir. 2020); electric car chargers, ChargePoint v. SemaConnect; garage door openers, Chamberlain Group v. Techtronic Industries; and digital cameras, Yu v. Apple (Fed. Cir. 2021), were considered abstract. In each case, the temptation to characterize the application of the tortured calculus of subject matter eligibility as having reached its nadir has been tempting, but the capacity for even greater flights of analytical fancy seems to have no limit. Sadly (for innovation, competitiveness, and the U.S. patent system), we have come very far from the simple rubric that "anything under the sun made by man" as a rational workable, subject matter eligibility standard.
PureCircle USA Inc. v. SweeGen, Inc. (Fed. Cir. 2024)
Nonprecedential disposition
Panel: Circuit Judges Dyk, Schall, and Stark
Opinion by Circuit Judge Dyk
* Those four being subject matter eligibility, written description, enablement, and obviousness-type double patenting.

Leave a comment