By Kevin E. Noonan —
 The prevailing attitude in many quarters is that the Biologics Price Competition and Innovation Act (BPCIA) has not facilitated approval of drugs biosimilar to reference biologic drug products with sufficient alacrity and has not reduced biologic drug prices that was the hope and justification for its enactment. For example, on February 3rd, the Federal Trade Commission and Food and Drug Administration issued a Joint Statement aimed at advancing competition in the biologic drug market. Earlier, the FDA itself had issued a Biosimilars Action Plan (BAP) providing specific goals to accelerate biosimilars competition, and the Trump Administration was reported to be considering a reduction of the 12-year exclusivity period for reference biologic drug products. Congress has not been silent (albeit has not passed any biologic drug legislation either), its members introducing bills such as the Biologic Patent Transparency Act (see "A Solution in Search of a Problem") and raising questions regarding settlements between biologic drug suppliers and biosimilar drug makers (see "Senators Ask FTC to Investigate Biosimilar Litigation Settlement Agreement").
The prevailing attitude in many quarters is that the Biologics Price Competition and Innovation Act (BPCIA) has not facilitated approval of drugs biosimilar to reference biologic drug products with sufficient alacrity and has not reduced biologic drug prices that was the hope and justification for its enactment. For example, on February 3rd, the Federal Trade Commission and Food and Drug Administration issued a Joint Statement aimed at advancing competition in the biologic drug market. Earlier, the FDA itself had issued a Biosimilars Action Plan (BAP) providing specific goals to accelerate biosimilars competition, and the Trump Administration was reported to be considering a reduction of the 12-year exclusivity period for reference biologic drug products. Congress has not been silent (albeit has not passed any biologic drug legislation either), its members introducing bills such as the Biologic Patent Transparency Act (see "A Solution in Search of a Problem") and raising questions regarding settlements between biologic drug suppliers and biosimilar drug makers (see "Senators Ask FTC to Investigate Biosimilar Litigation Settlement Agreement").
With this backdrop the FDA recently released a synopsis of the cumulative state of biosimilar drug approval to date, amounting to 26 approved biosimilar drugs (click on table to enlarge):
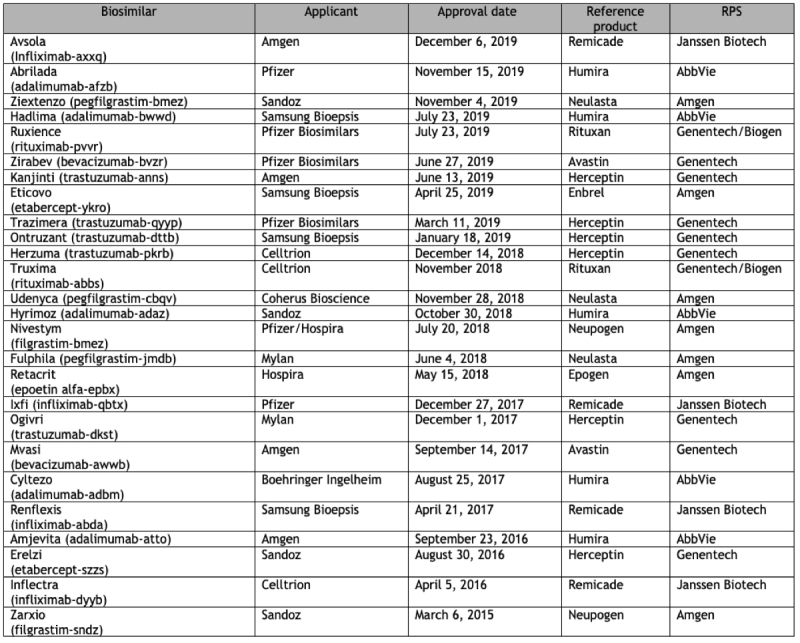
While encouraging, some sobering statistics are evident from this information. First, there have been a select few drugs that have been "targets" for competitors to make biosimilar versions, including Herceptin® (trastuzumab) (6 biosimilar versions); Humira® (5 biosimilar versions); Remicade® (infliximab) (4 biosimilar versions); Neupogen®/Neulasta® (filgrastim and pegfilgrastim, respectively) (2 and 3 biosimilar versions, respectively); Avastin® (bevacizumab) (2 biosimilar versions); Rituxan® (rituximab) (2 biosimilar version); Enbrel® (etabercept) (1 biosimilar version); and Epogen® (epoetin alfa) (1 biosimilar version). Part of the reason is that all of these drugs were approved prior to the enactment of the BPCIA and are outside the 12-year exclusivity period. In addition, these drugs were previously approved in Europe, and biosimilar applicants have benefited from FDA's decision to permit them to use in the U.S. comparative information and data between the reference product and the biosimilar submitted to the European authorities.
The biosimilar pipeline has 74 programs for 38 different reference products pending at the FDA, according to Dr. Sarah Yim, Acting Director of FDA's Office of Therapeutic Biologics and Biosimilars (although at least some of these are directed to the nine reference products for which FDA has already approved one or more biosimilars. This raises the question: at what point will the market saturate for any reference product, and will competition continue to decrease costs despite the belief that price reductions of more than 25-30% will be inhibited due to the expense of producing a biosimilar drug and obtaining regulatory approval? And other biologic drugs will (eventually) exhaust the 12 years of market exclusivity under the BPCIA). Despite Congressional and public impatience, the pace of biosimilar approval is increasing and this trend can be expected to continue.

Leave a comment