By Kevin E. Noonan –
In 2013, the U.S. Supreme Court rendered its decision in FTC v. Actavis, finding that although so-called reverse payment settlement agreements were not per se antitrust violations in cases brought against generic drug makers by under 35 U.S.C. § 271(e)(2), the potential for anticompetitive effects made them subject to antitrust scrutiny under the "rule of reason" standard. Four years on it is instructive to examine the effects of this decision, on the narrow issue of whether parties to ANDA litigation continue to enter into such settlements, and the broader question of whether generic drug products are entering the marketplace earlier that they would have without the Court's intervention.
 To recap, the case followed a long and tortuous series of cases where the Federal Trade Commission, in its own right or in support of private plaintiffs, attempted to convince one appellate court to differ from all the other Circuit Courts of Appeal, who had decided that no antitrust liability arose from such agreements so long as they were properly within the "scope of the patent." The opinions from the Second, Eleventh, and Federal Circuits were in agreement and provided detailed assessments of why such agreements were not anticompetitive, based on appropriate assertion of presumptively valid patent rights (as well as a recognition that in almost all of these cases the generic drug came to market earlier than if the patents had been litigated and found not invalid and infringed). The FTC finally prevailed in an action by private plaintiffs (In re K-Dur) appealed to the Third Circuit, where an appellate panel held these agreements to be anticompetitive under a "quick look" analysis (used for cases that, while not being per se illegal were sufficiently questionable that a complete rule-of-reason assessment was unwarranted). Although this decision gave the Commission the "circuit split" it needed to convince the Supreme Court to grant certiorari, the Court considered a companion case, brought by the Commission against Watson (later acquired by Actavis) and subject to a decision by the Eleventh Circuit adverse to the Commission's position. The Court agreed that this case presented a "superior vehicle" for its review inter alia because it had a more limited procedural history and was a matter of the actions of a Federal agency charged with policing antitrust activity and supported by that agency's expertise.
To recap, the case followed a long and tortuous series of cases where the Federal Trade Commission, in its own right or in support of private plaintiffs, attempted to convince one appellate court to differ from all the other Circuit Courts of Appeal, who had decided that no antitrust liability arose from such agreements so long as they were properly within the "scope of the patent." The opinions from the Second, Eleventh, and Federal Circuits were in agreement and provided detailed assessments of why such agreements were not anticompetitive, based on appropriate assertion of presumptively valid patent rights (as well as a recognition that in almost all of these cases the generic drug came to market earlier than if the patents had been litigated and found not invalid and infringed). The FTC finally prevailed in an action by private plaintiffs (In re K-Dur) appealed to the Third Circuit, where an appellate panel held these agreements to be anticompetitive under a "quick look" analysis (used for cases that, while not being per se illegal were sufficiently questionable that a complete rule-of-reason assessment was unwarranted). Although this decision gave the Commission the "circuit split" it needed to convince the Supreme Court to grant certiorari, the Court considered a companion case, brought by the Commission against Watson (later acquired by Actavis) and subject to a decision by the Eleventh Circuit adverse to the Commission's position. The Court agreed that this case presented a "superior vehicle" for its review inter alia because it had a more limited procedural history and was a matter of the actions of a Federal agency charged with policing antitrust activity and supported by that agency's expertise.
 The Supreme Court reversed the judgment below, in a decision by Justice Breyer supported by Justices Kennedy, Ginsburg, Sotomayor and Kagan; the Chief Justice dissented, joined by Justices Scalia and Thomas; Justice Alito recused himself from considering this case). Justice Breyer's majority opinion asserts that reverse payment settlement agreements can "sometimes violate the antitrust laws," and thus that the District Court should not have dismissed the case brought by the FTC. The opinion focused on the risk to the consuming public posed by such settlements in cases where the patent is invalid or not infringed. While the majority was willing to accept that the agreement's "anticompetitive effects fall within the scope of the exclusionary potential of the patent," this fact was not sufficient to "immunize the agreement from antitrust scrutiny." The majority's concern was that while the holder of a valid patent may be exempt from antitrust liability when enforcing the exclusionary right, ANDA litigation involves an allegation that either the patent is invalid (in which case the immunization is lost) or the generic product does not infringe (in which case the patent cannot be enforced against the non-infringing generic drug). Accordingly, the majority was convinced that such agreements "tend to have significant adverse effects on competition. The opinion states that "it would be incongruous to determine antitrust legality by measuring the settlement's anticompetitive effects solely against patent law policy, rather than by measuring them against procompetitive antitrust policies as well"; consequently:
The Supreme Court reversed the judgment below, in a decision by Justice Breyer supported by Justices Kennedy, Ginsburg, Sotomayor and Kagan; the Chief Justice dissented, joined by Justices Scalia and Thomas; Justice Alito recused himself from considering this case). Justice Breyer's majority opinion asserts that reverse payment settlement agreements can "sometimes violate the antitrust laws," and thus that the District Court should not have dismissed the case brought by the FTC. The opinion focused on the risk to the consuming public posed by such settlements in cases where the patent is invalid or not infringed. While the majority was willing to accept that the agreement's "anticompetitive effects fall within the scope of the exclusionary potential of the patent," this fact was not sufficient to "immunize the agreement from antitrust scrutiny." The majority's concern was that while the holder of a valid patent may be exempt from antitrust liability when enforcing the exclusionary right, ANDA litigation involves an allegation that either the patent is invalid (in which case the immunization is lost) or the generic product does not infringe (in which case the patent cannot be enforced against the non-infringing generic drug). Accordingly, the majority was convinced that such agreements "tend to have significant adverse effects on competition. The opinion states that "it would be incongruous to determine antitrust legality by measuring the settlement's anticompetitive effects solely against patent law policy, rather than by measuring them against procompetitive antitrust policies as well"; consequently:
[R]ather than measure the length or amount of a restriction solely against the length of the patent's term or its earning potential, as the Court of Appeals apparently did here, this Court answered the antitrust question by considering traditional antitrust factors such as likely anticompetitive effects, redeeming virtues, market power, and potentially offsetting legal considerations present in the circumstances, such as here those related to patents.
The Court also found that the "procompetitive" purposes of the Hatch-Waxman Act are consistent with having courts apply antitrust principles to reverse payment settlement agreements in ANDA litigation. The opinion enunciated five factors that lower courts should consider in applying the "rule of reason" to reverse payment settlement agreements, which amounted to 1) the size of the payment; 2) whether there are "legitimate justifications" for the agreement; 3) whether the patentee has "market power"; 4) the size of the reverse payment and whether it is "unexplained"; and 5) if there are other ways to settle, why do the parties choose a reverse payment. Importantly, the decision did not adopt the FTC's initial position (effectively disclaimed at oral argument) that these agreements should be presumptively unlawful or subject to the "quick look" analysis applied by the Third Circuit, because "the likelihood of a reverse payment bringing about anticompetitive effects depends upon its size, its scale in relation to the payer's anticipated future litigation costs, its independence from other services for which it might represent payment, and the lack of any other convincing justification."
However, these instructions left much of the work of deciding the quantum of evidence and scope of proof necessary for a court to make an antitrust determination to the lower courts. The value and extent of the majority's teachings on this question drew the Chief Justice's disdain in dissent, wherein he wrote "[g]ood luck to the district courts that must, when faced with a patent settlement, weigh the 'likely anticompetitive effects, redeeming virtues, market power, and potentially offsetting legal considerations present in the circumstances.'"
With this as background, it is instructive to review how the district courts and some appellate courts have grappled with the task given them (over the Chief Justice's misgivings) by the Actavis majority. Generally (and in anticipation of the Supreme Court's decision), settlements were crafted to avoid bald reverse payments in favor of non-monetary considerations. These include terms of such agreements where the branded company agreed not to produce an "authorized generic" version of a branded drug, or entering into supply agreements with the generic drug maker for active pharmaceutical ingredient (API) manufacturing, or licensing other, unrelated patents. These gambits yielded variable results for various challenges, either by the FTC or by consumer complaints (often brought by wholesale or resale pharmacies or other drug suppliers, or unions or other benefits providers.
The FTC has provided consolidated evidence and reports on these results; overall the number of ANDA settlements containing reverse payment terms has decreased by about 50% since the Actavis decision, with the trend being more prevalent for first ANDA filers. There has also been a reduction in the number of settlements involving first filers containing agreements by the branded drug maker not to market an "authorized generic" in competition with the generic entrant. The FTC Report reveals that 81-87% of ANDA litigation settlement agreements filed in FY 2014 did not contain any compensation from the branded to the generic company and/or restrictions on generic market entry.
In the courts, there is general recognition that both "extreme" positions were rejected by the Supreme Court; the patent grant does not give blanket immunity to antitrust liability, but the existence of the agreement does not presume liability either. In applying the "rule of reason," courts have come to different conclusions and used different standards (resulting in the unpredictability the Chief Justice foresaw). One of the first questions addressed has been whether the Court's decision limits antitrust scrutiny to those agreements containing payments of money. One case that addressed this question was In re Lipitor Antitrust Litigation (D. N.J. Sept. 12, 2014), where the District Court ruled that the Actavis standard is not limited to money settlements. A "payment," according to the Court, could be anything having value, but even though settlement agreements not having monetary terms (classic "reverse payments") can satisfy the Actavis standard, plaintiffs must plead sufficient facts to establish the economic value of what a generic drug maker receives:
[W]here Plaintiffs rely on a non-monetary reverse payment of an inchoate claim, they must plead plausible facts including an estimate the monetary value of same so the Actavis rationale can be applied. . . . To meet this standard, Plaintiffs must stand in the shoes of the underlying parties at the time of the settlement, and determine an estimate of the monetary value of the settlement at that time.
In the Lipitor case the District Court dismissed on the pleadings; the mere existence of a settlement is not enough, according to the opinion, a plaintiff must plead sufficient facts to establish the economic value of what the generic drug maker received, so that benefit could be used according to the Supreme Court's Actavis scheme for applying the rule of reason to the parties' activities. In this regard, the developing consensus for bringing an antitrust case puts on the plaintiff the burden of showing an agreement falls within the scope of Supreme Court's factors that indicate a court should perform a "rule of reason" antitrust assessment, which then shifts the burden to the defendant (or, more typically, defendants) to show the pro-competitive features of the agreement. The ultimate burden of establishing an antitrust violation always remains on the plaintiff.
For its part, the FTC has continued to mount antitrust challenges to settlement agreements in ANDA litigation, with varying results. When successful, however, the penalties can be chilling: for example, in two recent cases (In re: Opana ER Antitrust Litigation (MDL) (N.D. Ill. 2017); In re: Lidoderm Antitrust Litigation (N.D. Cal. 2017)) the Commission required antitrust defendants to abstain from settlements containing no-authorized generic and other terms for 10 years. In another recent case, Teva was forced to disgorge $1.2 billion received as the result of settlement (Federal Trade Commission v. Cephalon Inc. (E.D. Pa 2016)).
Results of representative cases involving antitrust allegations as the result of reverse payment settlement agreements in ANDA cases post-Actavis are tabulated below (click on table to enlarge).
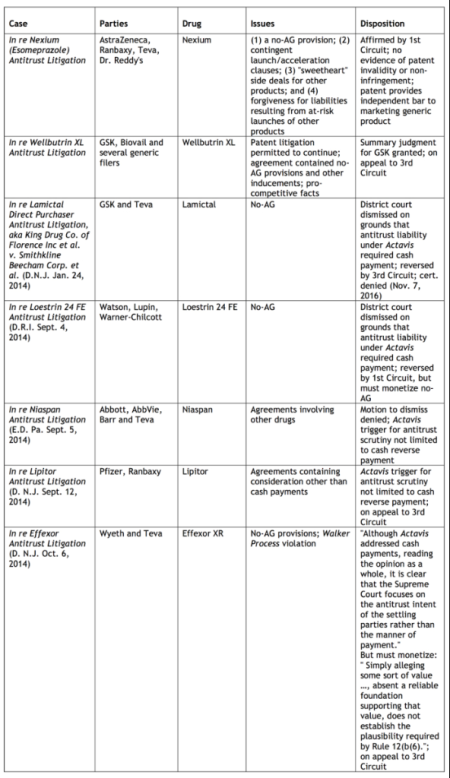
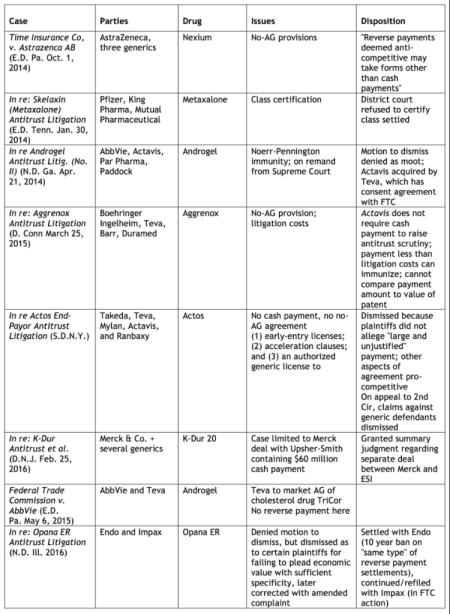
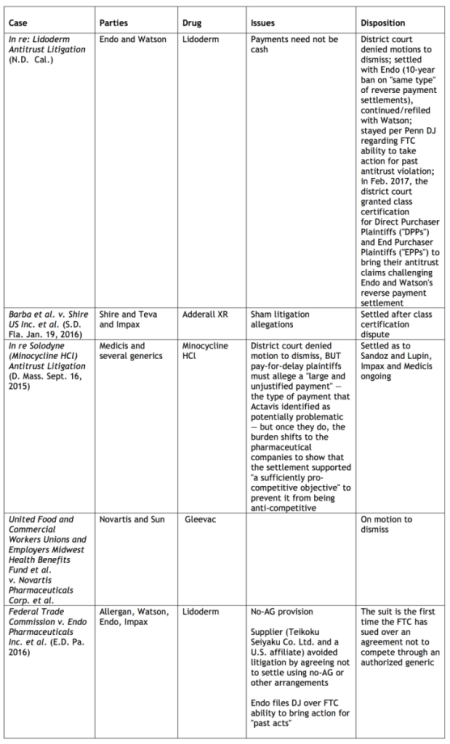

The economic importance of these issues is evident by the effect of generic drug competition on consumer prices for drugs. The General Accounting Office estimates U.S. prescription drug cost savings in excess of $1 trillion between 2003 and 2012, and the IMS Institute for Healthcare Informatics has accumulated statistics showing that generic drugs account for 29% of total drug sales and 86% of prescriptions. Duke economic Henry G. Grabowsky has published the results of his research showing that 70% of branded drug sales are lost to a generic version during the first month of generic entry into the marketplace, and that 84% of branded drugs are lost to their generic competitors within the first year of generic availability (Henry G. Grabowski et al., "Recent Trends in Brand-Name and Generic Drug Competition," Journal of Medical Economics, December 2013: 6–7).
More recently, another practice in the pharmaceutical industry termed "product hopping" has also raised antitrust scrutiny. This practice arises from FDA regulations regarding generic substitution: in order for a generic drug to be substituted by pharmacist, need branded counterpart (AB substitution). As the strategy has developed, a branded drug maker switches from one version of a drug to a related version and then clears pharmacy shelves of the earlier version. Consequently, a generic equivalent cannot take market share, because the branded drug maker has switched to a new version and there is no longer the possibility for AB substitution. This creates a tension between antitrust activities and competitive capitalist stratagems, between progress (sometimes a newer version really is "improved") and only cosmetic changes ("evergreening"). As these cases are turning out, whether antitrust liability lies may depend on whether branded drug maker acts to remove earlier version from pharmacy shelves. For example, in New York v. Actavis PLC et al. (2nd Cir. 2016), involving Namenda IR (newer ER version), efforts to remove the IR version from shelves were prevented by injunction. Similarly, contrast Mylan Pharmaceuticals Inc. et al. v. Warner Chilcott PLC et al. (E.D. Pa. 2015) (involving the drug Doryx) and In re: Suboxone Antitrust Litigation (E.D. Pa. 2015), where the drug in each case was pulled from pharmacy shelves, resulting in liability, with Walgreen Co. et al. v. AstraZeneca Pharmaceuticals LP et al. (D.D.C. 2015), where the prior omeprazole formulation was not pulled from pharmacy shelves in favor of the Nexium® substitute.
In sum, non-monetary terms of settlement (including branded innovator companies agreeing not to market competing "authorized generics") are proving to be enough to raise antitrust concerns, with courts instead looking at the entirety of what each party gains and loses. Although noninfringement allegations in Paragraph IV suits most likely suggest an antitrust violation, antitrust plaintiffs (including the FTC) will need to provide sufficient nexus between agreement terms and anticompetitive outcomes to sufficiently plead and to establish anticompetitive consequences under the rule of reason to prevail.
Adapted from an MBHB webinar, "Antitrust Issues in ANDA Patent Litigation," presented May 24, 2017.

Leave a comment