By Cambria Alpha-Cobb* and Anthony D. Sabatelli** —
 If innovation drives progress, then 2014 was the year of progress! During the past year the U.S. Food and Drug Administration approved 41 new drugs. That is 14 more than 2013 and the highest number in 18 years! What spurred this increase? And what challenges do these new drugs face in the 2015 market?
If innovation drives progress, then 2014 was the year of progress! During the past year the U.S. Food and Drug Administration approved 41 new drugs. That is 14 more than 2013 and the highest number in 18 years! What spurred this increase? And what challenges do these new drugs face in the 2015 market?
The FDA publishes the drug approval data based on New Molecular Entities (NMEs) [1]. These drugs can be both the traditional small molecule drugs in addition to newer high-molecular weight biologics. For information on NMEs and how they are classified, see "The Scorecard- Fewer New Drug Approvals in 2013: What's in store for 2014?" [2] a previous blog post by Dr. Anthony Sabatelli. Using data published annually from the FDA, we plotted the number of approved NMEs since the 1940s [1][3], below. After a dramatic peak in 1996, our plot shows a steady decline in approvals, followed by very little turnaround throughout the first decade of the 21st century and then a slow recovery in the past few years. Not only does this 2014 spike continue a slow upward trend, but also the new generation of drugs it embodies is believed to represent key advances in new drug development, suggesting a more broad-based recovery [4].
One of the main changes to the drug pipeline has been the expansion of treatment options. Many of the breakthrough discoveries from the past year are diverging from the traditional small molecules into whole new technologies and therapies. Nearly 4 out of 10 of the approved NMEs in 2014 were for biologics, a dramatic increase from 1 in 10 biologic NMEs approved in 2005 [5]. The 2014 NMEs spanned 10 therapeutic areas, up from 8 last year, with infectious diseases dominating with 12 approvals [4]. Next in line were approvals for cancer treatments (8) and rare disease drugs (5), emphasizing the industry's focus on specialized products [6].
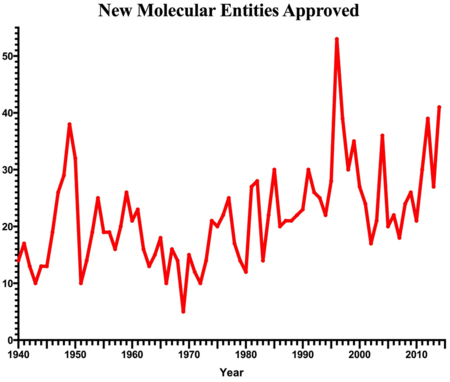 Chart of New Molecular Entities approved: Annual approved NMEs based on data from [1] and [3].
Chart of New Molecular Entities approved: Annual approved NMEs based on data from [1] and [3].
While this innovation is helping restore our faith in the drug industry, these drugs are arriving into a market where high prices are coming under attack, and more competition will only increase these attacks. Fierce Pharma identified six therapeutic areas, including some rare diseases and cancer treatment, where increased competition could lead to heavy price competition [7]. This competition raises questions about future commercial prospects and could suggest reduced future development efforts due to these new pricing pressures [8].
As John Carroll, editor of Fierce Biotech, states, "Approvals aren't enough to determine whether a pipeline strategy is successful or not." [8] However 2014 was the best year since 1996, and the innovative technologies provide encouragement that this spike might be part of a long-lived trend. Competition in pricing may hinder the sales of some of these new drugs, however the future is promising as firms venture from their comfort zones and embrace these breakthrough discoveries [8].
* Dr. Cambria Alpha-Cobb is a Technology Specialist at Dilworth IP
** Dr. Sabatelli is a Partner with Dilworth IP
[1]http://www.fda.gov/Drugs/DevelopmentApprovalProcess/DrugInnovation/ucm20025676.htm
[2]http://www.dilworthip.com/scorecard-fewer-new-drug-approvals-2013-whats-store-2014/
[3]http://www.fda.gov/AboutFDA/WhatWeDo/History/ProductRegulation/SummaryofNDAApprovalsReceipts1938tothepresent/default.htm
[4]http://www.forbes.com/sites/bernardmunos/2015/01/02/the-fda-approvals-of-2014/
[5]http://www.fda.gov/Drugs/DevelopmentApprovalProcess/HowDrugsareDevelopedandApproved/DrugandBiologicApprovalReports/NDAandBLAApprovalReports/ucm373420.htm
[6]http://in.reuters.com/article/2015/01/01/pharmaceuticals-approvals-idINL6N0UE2C120150101
[7]http://www.fiercepharma.com/story/limited-attention-span-focus-these-market-shake-ups-2015/2014-12-24
[8]http://www.fiercebiotech.com/special-reports/biopharma-posts-chart-topping-41-new-drug-approvals-2014/2015-01-02?utm_medium=nl&utm_source=internal

Leave a comment