By Donald Zuhn –-
 Last week, the Federal Circuit affirmed the rejection by the U.S. Patent and Trademark Office's Patent Trial and Appeal Board of claims 1-7 of U.S. Patent No. 6,284,471 as being unpatentable under the doctrine of obviousness-type double patenting. Janssen Biotech, Inc. and New York University ("Janssen"), co-assignees of the '471 patent, argued on appeal that obviousness-type double patenting was not applicable because the safe-harbor provision of 35 U.S.C. § 121 protected the '471 patent claims. In affirming the Board's decision, however, the Federal Circuit determined that the '471 patent was not entitled to safe-harbor protections.
Last week, the Federal Circuit affirmed the rejection by the U.S. Patent and Trademark Office's Patent Trial and Appeal Board of claims 1-7 of U.S. Patent No. 6,284,471 as being unpatentable under the doctrine of obviousness-type double patenting. Janssen Biotech, Inc. and New York University ("Janssen"), co-assignees of the '471 patent, argued on appeal that obviousness-type double patenting was not applicable because the safe-harbor provision of 35 U.S.C. § 121 protected the '471 patent claims. In affirming the Board's decision, however, the Federal Circuit determined that the '471 patent was not entitled to safe-harbor protections.
The '471 patent issued from U.S. Application No. 08/192,093, which was filed as a continuation-in-part application of U.S. Application Nos. 08/013,413 and 08/010,406. On the same day that the '093 application was filed, Janssen also filed U.S. Application No. 08/192,102, which was also filed as a continuation-in-part application of the '413 and '406 applications, and which later issued as U.S. Patent No. 5,656,272. Janssen subsequently filed U.S. Application No. 08/324,799, which was filed as a continuation-in-part of the '093 and '102 applications, and which later issued as U.S. Patent No. 5,698,185. The relationships of the above applications and patents is shown in the chart below, which is reproduced from the Federal Circuit's opinion.
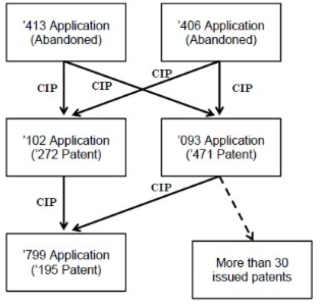
During prosecution of the '413 application, which relates to antibodies specific to human tumor necrosis factor alpha and included claims directed to a chimeric antibody and methods of treatment, a restriction requirement was issued in which Group I was directed to antibodies, pharmaceutical compositions, and assay methods, and Group IV was directed to methods for treating an animal by administering a pharmaceutical composition containing an antibody. Instead of replying to the restriction requirement, Janssen expressly abandoned the '413 application and filed the '093 and '102 applications as continuation-in-part applications of the '413 and '406 applications.
The '093 application disclosed and claimed subject matter from both the '413 and '406 applications (the '406 application relates to immunoreceptor molecules that are specific for TNF alpha or beta). After the '093 application was filed, Janssen allowed the '406 application to go abandoned. Janssen then filed a preliminary amendment in the '093 application, cancelling method claims designated as the invention of Group IV in the restriction requirement issued in the '413 application. However, Janssen did not limit the claimed subject matter to only that claimed and disclosed in the '413 application, as the '093 application still included claims directed to chimeric antibodies (based on the '413 application) and immunoreceptor molecules (based on the '406 application), and also retained language regarding binding to the TNF genus generally as opposed to the species of TNF alpha. In an Office action issued during prosecution of the '093 application, the claims were provisionally rejected on obviousness-type double patenting grounds over the '799 application. Janssen responded by amending the claims, including limiting claim 1 to TNF alpha. Janssen also filed preliminary amendments in both the '799 and '102 applications, replacing the pending claims of the '799 application with claims directed to methods of treating rheumatoid arthritis, and the pending claims of the '102 application with claims directed to methods of treating Crohn's disease.
Following the issuance of another Office action in the '093 application, in which the double patenting rejection was maintained, Janssen responded by arguing for withdrawal of the rejection in view of the amendment of the claims of the '799 application to recite methods of treating rheumatoid arthritis and by pointing to the restriction requirement issued in the '413 application, which Janssen argued precluded an obviousness-type double patenting rejection in the '093 application. The Examiner withdrew the double patenting rejection, and the '093 application eventually issued as the '471 patent, with claims directed to a chimeric antibody specific for TNF alpha (claims 1, 3, and 5–6), immunoassay methods for detecting human TNF (claims 2 and 4), and polypeptides of particular amino acid sequences that bind to human TNF alpha (claims 8 and 9).
Twelve years after the '471 patent issued, the USPTO instituted a reexamination of the patent (in response to a third-party request) on double patenting grounds over three patents including the '272 and '195 patents. During the reexamination, Janssen cancelled claims 8 and 9; requested that the benefit claim to the '406 application be deleted; sought amendment of the specification, abstract, and drawings to delete portions that were not in the '413 application; and requested that the application be designated as a divisional of the '413 application. The Examiner, however, maintained the double patenting rejections on the basis that the safe harbor did not apply, noting that more than thirty-two issued patents "reached through the '471 patent for benefit of a prior filing date" and that the "patentability of those claims . . . cannot be determined without reopening examination of those patents in view of the deletion of the subject matter in the '471 patent." The Examiner also determined that the one-way test for double patenting applied because the PTO was not "solely responsible" for the '471 patent's later issuance, and that the claims of the '471 patent were unpatentable under that test. The Board subsequently affirmed the Examiner's determination, and Janssen appealed the Board's decision to the Federal Circuit.
In an opinion authored by Chief Judge Prost, and joined by Circuit Judges Reyna and Wallach, the Federal Circuit noted that "[t]he main issue on appeal is whether the safe-harbor provision of 35 U.S.C. § 121 applies to the '471 patent and protects it from invalidation based on the '272 and '195 reference patents." The opinion also noted that "[t]he § 121 safe harbor, 'by its literal terms, protects only divisional applications (or the original application) and patents issued on such applications,'" quoting Pfizer, Inc. v. Teva Pharm. USA, Inc., 518 F.3d 1353, 1360 (Fed. Cir. 2008), and explained that:
[P]atents issued on CIP applications are not within the scope of § 121. [Pfizer, 518 F.3d] at 1362. Nor are patents issued on continuation applications. [Amgen Inc. v. F. Hoffman-La Roche Ltd., 580 F.3d 1340, 1354 (Fed. Cir. 2009)]. Our precedent is clear: aside from the original application and the original patent, the protection afforded by § 121 is limited to divisional applications and patents issued on divisional applications. Pfizer, 518 F.3d at 1362.
The panel noted that in G.D. Searle LLC v. Lupin Pharm., Inc., 790 F.3d 1349 (Fed. Cir. 2015), the question of whether a patent owner can retroactively bring a challenged patent, issued from a CIP application, within the scope of the § 121 safe harbor by amending the CIP application after issuance to redesignate the application as a divisional application was answered by the Federal Circuit in the reissue context, where the Court determined that the patent owner could not take advantage of the safe-harbor provision simply by designating the CIP as a divisional application. Applying Searle to the instant case, the panel indicated that it was "persuaded by the reasoning in Searle that a patent owner cannot retroactively bring its challenged patent within the scope of the safe-harbor provision by amendment in a reexamination proceeding," stating that "once the '471 patent issued on the '093 application—which, like the application in Searle, at the time of issuance included new matter not disclosed in the original application and so was a properly designated CIP—the '471 patent was barred from safe-harbor protections."
The panel pointed out that "[t]he '471 patent cannot retroactively become, for the purposes of § 121, a 'patent issued on' a divisional application after it already issued on a CIP application; not even if that CIP application is effectively redesignated as a divisional application during reexamination," and noted that "[f]or a challenged patent to receive safe-harbor protections, the application must be properly designated as a divisional application, at the very latest, by the time the challenged patent issues on that application." In response to Janssen's argument that it had not enjoyed a benefit from having filed the '093 application as a CIP of the '413 and '406 applications because no issued claims in the '471 patent relied upon the new matter in the '093 application, the panel disagreed, explaining that "the examiner found that Janssen had benefitted because more than thirty patents issued to Janssen claiming priority to the '471 patent and/or the '093 application as a CIP of both the '406 and '413 applications."
The opinion notes that given the panel's determination that once the '471 patent had issued on a CIP application, it was not entitled to safe-harbor protections of § 121, the panel did not need to consider the USPTO's alternative argument that § 121 does not apply because the '471 patent and the reference patents did not maintain consonance with the restriction requirement made in the '413 application. In addition, in response to Janssen's argument that the Federal Circuit's holding "creates a rigid 'divisional as filed' test," and ignores longstanding USPTO rules and practices that permit applicants to amend the disclosure and claims of an application after it is filed and to change an application's relationship to a prior-filed application, the panel noted that "[t]hese practices, whether or not they are widely practiced or even permitted under PTO rules, have no relevance here because Janssen did not follow them." The panel also explained that:
[W]e do not decide whether such filing practices or amendments made prior to issuance—wherein an application is designated as a divisional application by the time the challenged patent issues on that application—would be sufficient to bring the challenged patent within the scope of the safe-harbor protections.
The opinion concludes with the panel finding to be unpersuasive Janssen's argument that the Board erred by failing to apply a two-way test for double patenting. Instead, the panel concluded that Janssen failed to establish that the PTO is "solely responsible" for any alleged delay associated with the issuance of the '471 patent. Finding that the '471 patent was not entitled to safe-harbor protections, and that the Board did not err in applying the one-way test for double patenting, the Federal Circuit affirmed the Board's rejection of claims 1–7 of the '471 patent as being unpatentable under the doctrine of obviousness-type double patenting.
In re Janssen Biotech, Inc. (Fed. Cir. 2018)
Panel: Chief Judge Prost and Circuit Judges Reyna and Wallach
Opinion by Chief Judge Prost

Leave a comment