By Andrew Williams —
 Earlier today, both parties to the AbbVie v. Amgen litigation announced a settlement that resolves all intellectual property-related litigation over Amgen's FDA-approved adalimumab biosimilar AMGEVITA™/AMJEVITA™ (see AbbVie press release & Amgen press release). While the financial terms of the agreement were not disclosed, it was reported that AbbVie will grant non-exclusive patent licenses worldwide, on a country-by-country basis, for the use and sale of Amgen's biosimilar product. AMGEVITA is expected to launch in most countries of the European Union on October 16, 2018, and AMJEVITA (adalimumab-atto) in the U.S. on January 31, 2023. AbbVie reported that all related pending litigation will be dismissed, and that "Amgen has acknowledged the validity of AbbVie's intellectual property related to HUMIRA." The prices of both companies' stock were up at the end of the day, although on a percentage basis, AbbVie's was up higher. This could be due to the certainty in the delay of competition that AbbVie will face from Amgen in the United States. However, HUMIRA could face competition from Boehringer Ingelheim's biosimilar Cyltezo (adalimumab-adbm), which was approved by the FDA in late August (see "FDA Approves First Cancer-Treatment Biosimilar — Amgen's Mvasi").
Earlier today, both parties to the AbbVie v. Amgen litigation announced a settlement that resolves all intellectual property-related litigation over Amgen's FDA-approved adalimumab biosimilar AMGEVITA™/AMJEVITA™ (see AbbVie press release & Amgen press release). While the financial terms of the agreement were not disclosed, it was reported that AbbVie will grant non-exclusive patent licenses worldwide, on a country-by-country basis, for the use and sale of Amgen's biosimilar product. AMGEVITA is expected to launch in most countries of the European Union on October 16, 2018, and AMJEVITA (adalimumab-atto) in the U.S. on January 31, 2023. AbbVie reported that all related pending litigation will be dismissed, and that "Amgen has acknowledged the validity of AbbVie's intellectual property related to HUMIRA." The prices of both companies' stock were up at the end of the day, although on a percentage basis, AbbVie's was up higher. This could be due to the certainty in the delay of competition that AbbVie will face from Amgen in the United States. However, HUMIRA could face competition from Boehringer Ingelheim's biosimilar Cyltezo (adalimumab-adbm), which was approved by the FDA in late August (see "FDA Approves First Cancer-Treatment Biosimilar — Amgen's Mvasi").
 As we reported at the time, AbbVie filed suit against Amgen in the United States District Court for the District of Delaware on August 4, 2016, pursuant to the BPCIA and 35 U.S.C. 271(e)(2)(C). In its Counterclaims and Answer, Amgen reported that it had certified to AbbVie that it would not begin commercial marketing of its biosimilar before at least one of the patents identified by AbbVie had expired on December 31, 2016. Obviously, Amgen did not launch-at-risk after that date. Moreover, Amgen had stated that it intended to fully comply with the 180-day Notice of Commercial Marketing provision of the BPCIA (at least as it understood it before the Supreme Court's Sandoz v. Amgen case), which would have prevented them from launching until at least March, 2017. However, the present litigation only encompassed 10 of the 61 patents identified by AbbVie and Amgen during the so-called patent dance. Because AbbVie would have certainly filed the second-phase litigation if Amgen had provided commercial-marketing notice, it is reasonable to assume that such notice was not yet given.
As we reported at the time, AbbVie filed suit against Amgen in the United States District Court for the District of Delaware on August 4, 2016, pursuant to the BPCIA and 35 U.S.C. 271(e)(2)(C). In its Counterclaims and Answer, Amgen reported that it had certified to AbbVie that it would not begin commercial marketing of its biosimilar before at least one of the patents identified by AbbVie had expired on December 31, 2016. Obviously, Amgen did not launch-at-risk after that date. Moreover, Amgen had stated that it intended to fully comply with the 180-day Notice of Commercial Marketing provision of the BPCIA (at least as it understood it before the Supreme Court's Sandoz v. Amgen case), which would have prevented them from launching until at least March, 2017. However, the present litigation only encompassed 10 of the 61 patents identified by AbbVie and Amgen during the so-called patent dance. Because AbbVie would have certainly filed the second-phase litigation if Amgen had provided commercial-marketing notice, it is reasonable to assume that such notice was not yet given.
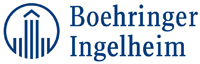 The AbbVie v. Boehringer Ingelheim litigation, on the other hand, is just getting started. On August 2, 2016, almost a year after the Amgen case, AbbVie sued Boehringer pursuant to the BPCIA and 35 U.S.C. 271(e)(2)(C) because Boehringer submitted its application to the FDA to market its HUMIRA® biosimilar. Boehringer answered the Complaint on September 11, 2017. AbbVie alleged that there are more than 100 patents concerning the HUMIRA product, and that it identified 74 of which that were (or would be) infringed. Nevertheless, because Boehringer, as the Biosimilar application, had the ability to cap the number of patents in the suit, the initial phase litigation only concerns eight patents. Boehringer had selected the number of patents each side could identify for litigation as five, but the litigation only concerns eight patents because two appeared on both lists. The second wave can only occur after Boehringer provides its 180-day commercial-marketing notice.
The AbbVie v. Boehringer Ingelheim litigation, on the other hand, is just getting started. On August 2, 2016, almost a year after the Amgen case, AbbVie sued Boehringer pursuant to the BPCIA and 35 U.S.C. 271(e)(2)(C) because Boehringer submitted its application to the FDA to market its HUMIRA® biosimilar. Boehringer answered the Complaint on September 11, 2017. AbbVie alleged that there are more than 100 patents concerning the HUMIRA product, and that it identified 74 of which that were (or would be) infringed. Nevertheless, because Boehringer, as the Biosimilar application, had the ability to cap the number of patents in the suit, the initial phase litigation only concerns eight patents. Boehringer had selected the number of patents each side could identify for litigation as five, but the litigation only concerns eight patents because two appeared on both lists. The second wave can only occur after Boehringer provides its 180-day commercial-marketing notice.
Specifically, for the first wave, AbbVie identified:
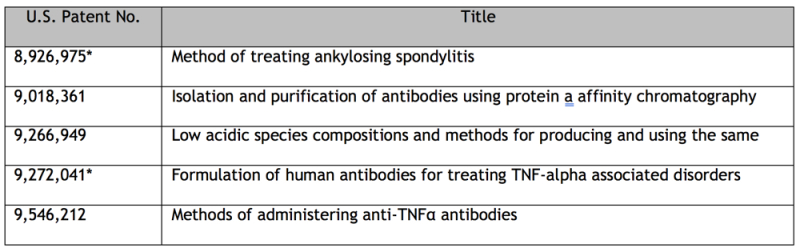
One of these, the '041 patent, is the same as it asserted in the Amgen litigation. For its part, Boehringer identified (with the common patents identified by the symbol "*"):
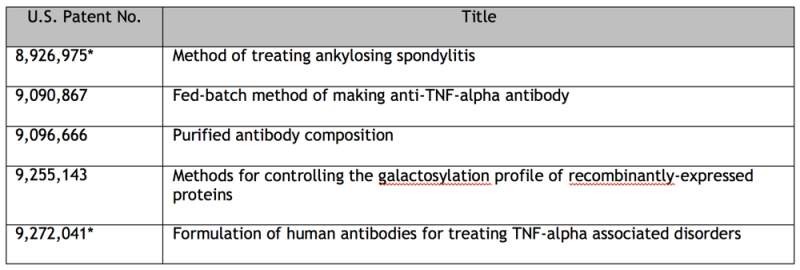
Boehringer also identified AbbVie's '041 patent asserted in the Amgen case, and listed the '666 patent, which Amgen had similarly listed. For those interested, the entire list of 74 patents can be found on pages 15-19 of the complaint. Boehringer asserted, though, that at least four of these patents were improperly listed, as they had either expired or had been determined by the PTAB to be unpatentable. This case is at the very early stages, and there have been no reports of its settlement. As always, we will continue to monitor this case and provide updates as warranted.

Leave a comment