Method for Manufacturing Product (Dental Crown) Found to be Patent Eligible
By Joseph Herndon —
 Zircore sued Straumann Manufacturing for patent infringement in the U.S. District Court for the Eastern District of Texas (Marshall Division) of three patents including U.S. Patent No. 7,967,606. Straumann moved to dismiss Zircore's allegations with respect to the '606 patent for failure to state a claim, contending that the '606 patent claims are not patent eligible under 35 U.S.C. § 101. The Court denied the motion and found that the '606 patent was not directed to an abstract idea, but alternatively, was directed to a method of manufacturing physical crown copings for prosthodontics. This was found to be necessarily rooted in the physical world.
Zircore sued Straumann Manufacturing for patent infringement in the U.S. District Court for the Eastern District of Texas (Marshall Division) of three patents including U.S. Patent No. 7,967,606. Straumann moved to dismiss Zircore's allegations with respect to the '606 patent for failure to state a claim, contending that the '606 patent claims are not patent eligible under 35 U.S.C. § 101. The Court denied the motion and found that the '606 patent was not directed to an abstract idea, but alternatively, was directed to a method of manufacturing physical crown copings for prosthodontics. This was found to be necessarily rooted in the physical world.
The '606 Patent
When a patient requires a tooth replacement, a three-dimensional model of the patient's mouth is prepared. Using the model of the patient's mouth, a lab technician will fit a metal portion of an abutment into an appropriate location in the model. The model will then be scanned. The scan provides data about an orientation of a metal insert within the model of the mouth and also data about the existing teeth surrounding the position of the abutment. The data from the scan, along with stored data about the standard shape of the metal insert to which the ceramic portion is to be mounted, is used to determine and design the appropriate shape for the ceramic portion of the abutment. At the same time, the shape of the coping is internally designed, using the data so that the coping will fit over the designed ceramic portion of the abutment.
The invention includes designing the coping and infrastructure by first determining the shape and orientation of the final crown, subtracting a thickness for the crown from the shape to determine the shape and orientation of a coping, and subtracting a thickness for the coping to form the shape and orientation of an abutment.
The invention allows both the coping at the ceramic portion of the abutment to be manufactured from a single block, significantly reducing the amount of material required. In addition, the inventive system significantly reduces the amount of labor necessary to manufacture the coping and the abutment. Because the abutment is custom manufactured, a superior mounting surface is achieved, regardless of the orientation of the metal insert with the mouth.
Figure 2 of the '606 patent is reproduced below, and diagrammatically illustrates the manufacturing process.
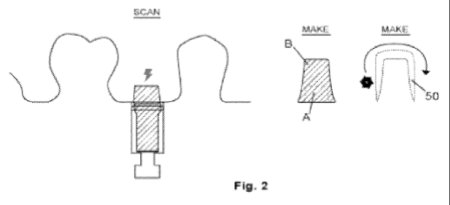
Claim 1 is representative and reproduced below:
1. A method of manufacturing custom crown copings and infrastructures comprising the steps of:
preparing a three-dimensional model of a patient's mouth;
fitting an implant abutment insert into the model;
storing data about the implant abutment insert standard size and shape in a file;
scanning the model while the implant abutment insert is in the model;
utilizing the implant abutment insert size data and data from said scanning step to determine and design a core to fit over the insert and at the same time determine and design a coping to fit over the core; and
manufacturing said core and said coping.
Patent Eligibility
In assessing subject matter eligibility, a court must first determine whether the claims at issue are directed to a patent ineligible concept. If the claims are directed to an ineligible concept, the court must then consider the elements of each claim both individually and as an ordered combination to determine whether the additional elements transform the nature of the claim into a patent eligible application.
Straumann alleged that the claims are directed to the abstract idea of designing two components at the same time. This highly generic view of the claims was rejected by the Court.
The Court found that Straumann isolated two steps from the '606 patent claims, the combination of which is allegedly the point of novelty, and ignored the remainder of the claims. Contrary to Straumann's (Defendants) assertion, the '606 patent claims are directed to a method of manufacturing physical crown copings for prosthodontics.
The '606 patent describes a physical process for collecting information about a patient's mouth, preparing a three-dimensional model of the mouth, scanning the model, and on the basis of data collected from this process, manufacturing the custom crown coping. The Court found that the context of the patent as a whole suggests that the invention is rooted in the physical world.
The Defendants suggested that the claims, at least on their face, appear to include certain steps that could be construed as computerized steps, and thus steps that could arguably be carried out mentally. But the Defendants cited no authority for the proposition that a claim reciting a method of manufacturing becomes abstract simply because it includes modeling or involves the use of computerized data.
The Court found that the '606 patent claims look nothing like the claims found to be abstract in previous cases, including claims within the two major abstract categories — those directed solely to collecting and analyzing information, and those involving fundamental economic and conventional business practices.
The Court appeared to take a realistic and pragmatic view of the claim, namely, that it is directed to manufacturing custom crown copings and infrastructures, which is necessarily rooted in in the physical world.
Accordingly, because the Court found that the '606 patent claims are not directed to an abstract idea, the Court found that Zircore had plausibly stated a claim for patent infringement and the motion was denied.
Report and Recommendation by Magistrate Judge Roy S. Payne

Leave a comment