By Kevin E. Noonan —
 At the end of March, the Intellectual Property Counsels' Committee (IPCC) of the Biotechnology Innovation Organization (BIO) held its Spring meeting, convening in-house counsel and outside counsel involved in patenting the member companies' intellectual property. Presenting on one of the panels was Professor Jacob S. Sherkow (at right) from the New York Law School's Innovation Center for Law and Technology; his presentation provided the most recent statistics on the activities of the Patent Trial and Appeal Board related to inter partes reviews (IPR).
At the end of March, the Intellectual Property Counsels' Committee (IPCC) of the Biotechnology Innovation Organization (BIO) held its Spring meeting, convening in-house counsel and outside counsel involved in patenting the member companies' intellectual property. Presenting on one of the panels was Professor Jacob S. Sherkow (at right) from the New York Law School's Innovation Center for Law and Technology; his presentation provided the most recent statistics on the activities of the Patent Trial and Appeal Board related to inter partes reviews (IPR).
According to Professor Sherkow, there have been 4,253 IPR petitions filed; only 228 (5.36%) of these have been on Orange Book-listed patents. Looking more closely at these 228 petitions:
• 152 (66.67%) were filed by generic manufacturers;
• 37 (16.22%) were filed by brand manufacturers;
• 37 (16.22%) were filed by investment firms, and
• 2 (0.88%) were filed by public interest organizations.
The most prodigious generic filer has been Mylan (filing 22), followed by Lupin (16), Apotex (16), Amneal (16), Par (11), and Praxair (10). These statistics illustrate how the IPR strategy has been inculcated into the Hatch-Waxman regime for challenging patents and can provide an alternative route to litigation for challenging Orange-Book-listed patents.
Looking at outcomes, 98 (42.9%) of these IPRs were instituted and 68 (29.8%) not instituted, with 52 (22.8%) having institution decisions pending and 10 (4.3%) being settled before institution. Of the 98 that have been instituted, most (66) are awaiting trial, while 12 settled, 12 were found in final written decision to have patentable claims and 5 had the Board determine that all claims were unpatentable. These statistics suggest that while providing a low(er)-cost alternative to Hatch-Waxman litigation there has not been wholesale invalidation of these Orange Book-listed patents such as was seen for the initial cadre of IPRs (which were invalidated with such numbing consistency that the Board was termed a "death panel" for patents).
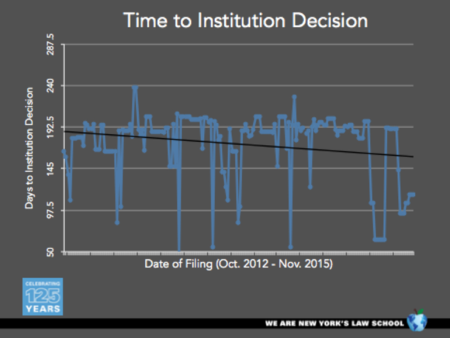
Professor Sherkow also showed graphs plotting the time the Board took to make its institution decision between October 2012 and November 2015:
and the rate of petition filings during that timeframe:
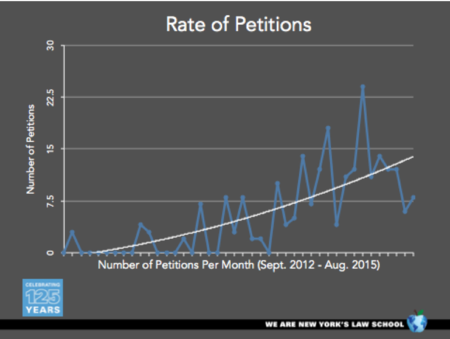
These graphs show that the institution times have been variable but decreasing generally, from almost six months for the first-filed IPRs to a little less that 5 months currently, and that the rate of petition filing has increased from around five per month to about fifteen per month by August 2015.
The IPR option has received a fair amount of attention since the Office implemented the process as part of its rollout of the America Invents Act in 2012. It should be remembered that the predecessor to these petitions, petitions for inter partes reexamination established in 1999 by the American Inventor Protection Act, took some time to be used (but not to the extent that IPRs have been used almost from their institution). The "threat" of estoppel in IPR use apparently has been countered by petitioners' early successes (and success rate) before the Board, and by the procedural advantages that have been given to the petitioner (including using the broadest reasonable interpretation, or BRI, of the claims coupled with impediments raised against the patentee regarding entering claim amendments). But insofar as the USPTO can view the level of petitions filed against granted U.S. patents to be a success, these statistics support that interpretation. Whether IPRs will ultimately help innovation or hinder it is another, as yet unanswered question.

Leave a comment