By Kevin E. Noonan —
That it is more difficult today to be a patentee able to defend her patent rights than any time since the 1940's is nicely illustrated by the Federal Circuit's decision in Par Pharmaceutical, Inc. v. TWI Pharmaceuticals, Inc., where the patentee prevailed only to face almost certain defeat on remand.
 This is an ANDA case involving certain formulations of megestrol acetate, a compound known in the art as an appetite stimulant for use with cancer patients and AIDS patients to treat cachexia and for treating anorexia and other conditions where there is a significant loss of body mass. The invention provided modified megestrol formulations where the drug was administered as "nanosized" particles; the prior art administered megestrol acetate in micronized form, sold by Bristol-Myers Squibb as Megace OS, which formulation had been the target of ANDA litigation. Par had obtained FDA approval for marketing its own form of Megace OS, but rather than enter the marketplace with this formulation contracted with Alkermes Pharma Ireland (formerly Elan Pharmaceuticals) to adapt that company's nanoparticle technology (termed "NanoCrystals") to make nanosized megestrol acetate formulations (which it termed Megace ES and for which Par also received FDA approval).
This is an ANDA case involving certain formulations of megestrol acetate, a compound known in the art as an appetite stimulant for use with cancer patients and AIDS patients to treat cachexia and for treating anorexia and other conditions where there is a significant loss of body mass. The invention provided modified megestrol formulations where the drug was administered as "nanosized" particles; the prior art administered megestrol acetate in micronized form, sold by Bristol-Myers Squibb as Megace OS, which formulation had been the target of ANDA litigation. Par had obtained FDA approval for marketing its own form of Megace OS, but rather than enter the marketplace with this formulation contracted with Alkermes Pharma Ireland (formerly Elan Pharmaceuticals) to adapt that company's nanoparticle technology (termed "NanoCrystals") to make nanosized megestrol acetate formulations (which it termed Megace ES and for which Par also received FDA approval).
During prosecution, the PTO rejected method claims for using these formulations as being obvious over the combination of prior art disclosing micronized megestrol acetate formulations with prior art NanoCrystal formulation methods. The Office initially found these formulation claims obvious, but Par obtained U.S. Patent No. 7,101,576 by including "wherein" clauses, based on its discovery that the nanosized formulations had a reduced "food effect" when compared to micronized formulations; specifically, the nanosized version did not increase nutrient absorption (rate or extent) which the panel noted was important for treating AIDS patients. Claim 1 of the '576 patent is illustrative:
A method of increasing the body mass in a human patient suffering from anorexia, cachexia, or loss of body mass, comprising administering to the human patient a megestrol formulation, wherein:
(a) the megestrol acetate formulation is a dose of about 40 mg to about 800 mg in about a 5 mL dose of an oral suspension;
(b) the megestrol acetate formulation comprises megestrol particles having an effective average particle size of less than about 2000 nm, and at least one surface stabilizer associated with the surface of the megestrol particles; and
(c) the administration is once daily;
wherein after a single administration in a human subject of the formulation there is no substantial difference in the Cmax of megestrol when the formulation is administered to the subject in a fed versus a fasted state,
wherein fasted state is defined as the subject having no food within at least the previous 10 hours, and wherein fed state is defined as the subject having a high-calorie meal within approximately 30 minutes of dosing.
 TWi Pharmaceuticals filed an ANDA for Par's nanosized formulation, and in its Paragraph IV letter asserted invalidity on four grounds: "(1) the claims are invalid as obvious under 35 U.S.C. § 103 []; (2) the claims do not cover patentable subject matter under § 101; (3) the claims are not enabled under § 112; and (4) Par does not have standing to assert its claims."
TWi Pharmaceuticals filed an ANDA for Par's nanosized formulation, and in its Paragraph IV letter asserted invalidity on four grounds: "(1) the claims are invalid as obvious under 35 U.S.C. § 103 []; (2) the claims do not cover patentable subject matter under § 101; (3) the claims are not enabled under § 112; and (4) Par does not have standing to assert its claims."
The District Court did not reach any of TWi's invalidity contentions other than obviousness, for which it based its opinion on several prior art references that fell into two broad categories: first, art related to "the pharmacokinetic properties of megestrol, and [second] the use of nanoparticle technology in drug formulation." In addition to the prior art use of Megase OS, the Court considered scientific references relating to how the drug was absorbed using micronized formulations. With regard to nanoparticle technology used for drug formulations the District Court considered prior U.S. and EP patents showing that particles having sizes in the range of 400-1000 nm were useful particularly for drugs having low aqueous solubility. Included in these references was U.S. Patent No. 5,145,684 ("'684 patent"), which disclosed improved bioavailability of Class II drugs related to megestrol using nanosized technology. Another reference particularly noted by the District Court was a reference suggesting that nanosized drug formulations could be used to improve bioavailability and decrease the food effect, in R.H. Müller et al, Nanosuspensions as Particulate Drug Formulations in Therapy: Rationale for Development and What We Can Expect for the Future, 47 Advanced Drug Delivery Rev. 3–19 (2001); Par relied upon these advantages to overcome an obviousness rejection during patent prosecution.
While the District Court was unwilling to grant summary judgment of invalidity to TWi, it did enter judgment against Par after a bench trial, finding that the use of megestrol and the advantages of nanosized drug formulations were known in the prior art. To counter Par's argument that the prior art did not teach the advantageous properties of nanosized megestrol acetate formulations on the "food effect" as recited in the "wherein" clauses of Par's claims, the District Court held that these were the consequence of the "pharmacokinetic parameters of these formulations" and thus were "inherent" properties of these formulations. The Court also found that known patient-to-patient variabilities in prior art micronized megestrol formulations like Megace OS provided sufficient motivation to use nanosized formulations of megestrol even in the absence of any teachings in the art regarding the beneficial reduced food effect. Thus, the District Court found for TWi that Par's claims were obvious.
The Federal Circuit disagreed, vacating the judgment and remanding for further fact-finding, in an opinion by Judge O'Malley, joined by Judges Wallach and Hughes. After setting forth the standard of review and the law of obviousness generally, the panel set forth the dispositive issue in the case: whether all the claimed limitations were disclosed in the prior art (which the opinion sets out as a threshold issue that must be satisfied before questions regarding motivation to combine or reasonable expectation of success can be considered), citing Medichem, S.A. v. Rolabo, S.L., 437 F.3d 1157, 1164 (Fed. Cir. 2006). The parties generally agreed about the content of the prior art. In particular, it was also undisputed that the prior art did not expressly disclose any food effect associated with megestrol administration. The question was whether the food effect limitation was disclosed in the prior art under the principles of inherency. The District Court found that the food effect limitation was inherently disclosed in the art; the Federal Circuit held this was error.
The panel's analysis began with the definition of inherency and the established principle that "inherency may supply a missing claim limitation in an obviousness analysis," citing Santarus, Inc. v. Par Pharm., Inc., 694 F.3d 1344, 1354 (Fed. Cir. 2012); Alcon Research, Ltd. v. Apotex Inc., 687 F.3d 1362, 1369 (Fed. Cir. 2012); In re Kao, 639 F.3d 1057, 1070 (Fed. Cir. 2011); In re Kubin, 561 F.3d 1351, 1357 (Fed. Cir. 2009), and Ansonia Brass & Copper Co. v. Elec. Supply Co., 144 U.S. 11 (1892) (later in the opinion the panel notes that none of these cases have changed the fundamental approach to the question cited below). But the opinion reiterates the Court's caution that inherency cannot be based on what may happen but what must happen by a combination of prior art that purportedly will inherently have a property recited as an affirmative limitation in a claim being examined for obviousness, citing In re Rijckaert, 9 F.3d 1531, 1533–34 (Fed. Cir. 1993); In re Oelrich, 666 F.2d 578, 581 (C.C.P.A. 1981); and Application of Shetty, 566 F.2d 81, 86 (C.C.P.A. 1977). The principle at play in the Court's assessment of the District Court's decision is found in Hansgirg v. Kemmer, 102 F.2d 212, 214 (C.C.P.A. 1939):
Inherency, however, may not be established by probabilities or possibilities. The mere fact that a certain thing may result from a given set of circumstances is not sufficient. If, however, the disclosure is sufficient to show that the natural result flowing from the operation as taught would result in the performance of the questioned function, it seems to be well settled that the disclosure should be regarded as sufficient.
Here, the Federal Circuit found that the District Court did not require TWi to produce clear and convincing evidence of such inevitability. Indeed, the panel noted that the claims themselves contain limitations that contradict what the opinion calls the "district court's broad diktats regarding the effect of particle size on bioavailability and food effect." The panel acknowledged that it may be the case that reducing particle size is accompanied by a decrease in the food effect, but vacated the judgment because the lower court did not require TWi to provide clear and convincing evidence that such a correlation between decreasing particle size and decreased food effect must occur as required for an inherently disclosed element of a claim when evaluating it for obviousness. Thus the panel remanded, after further finding that the District Court had not erred in finding both a motivation to combine the cited art and a reasonable expectation of success in producing the claimed formulations. In coming to these conclusions the panel rejected Par's contention that the skilled worker would not have combined the art to achieve the claimed effect, reminding the parties (and us) that the motivation identified by a court need not be the same motivation that compelled the inventors, citing Allergan, Inc. v. Sandoz, Inc., 726 F.3d 1286, 1292 (Fed. Cir. 2013). This principle addressed Par's challenge that motivation to combine could not have been present because the art did not recognize the benefits of the combination with regard to the food effect discovered by the patentees. The Federal Circuit found several other motivations related to, inter alia, interpatient variability in the prior art that served it quite nicely in satisfying the requirement for a motivation to combine. And while Par argued that these motivations were better served by other modifications in prior art micronized megestrol formulations, the panel noted that "the motivation [need not] be the best option, only [] a suitable option from which the prior art did not teach away," citing Galderma Labs., L.P. v. Tolmar, Inc., 737 F.3d 731, 738 (Fed. Cir. 2013), and Bayer Healthcare Pharm., Inc. v. Watson Pharm., Inc., 713 F.3d 1369, 1376 (Fed. Cir. 2013).
Regarding the reasonable expectation of success, the opinion found that the NanoCrystal technology was sufficiently well established that the skilled worker would have expected to be able to adapt the prior art micronized formulations of megestrol acetate to the claimed nanosized formulations and that these would have a reasonable likelihood (albeit not a certainty, which is not required under the In re O'Farrell standard, 853 F.2d 894, 903–04 (Fed. Cir. 1988)) to be operable. The panel also rejected Par's contentions that the prior art taught away from micronized megestrol acetate formulations, particularly because that art did not discuss important features of the claimed invention (such as nanosized megestrol formulations). Finally, the opinion also rejected Par's evidence of secondary considerations.
Despite this victory on appeal, Par's problems are only beginning in view of TWi's assertion of an invalidity defense based on subject matter eligibility. In addition to being several district courts' basis du jour for invalidating claims, the similarities between the claims at issue in this case and the Prometheus claims struck down by the Supreme Court in Mayo v. Prometheus is uncanny:
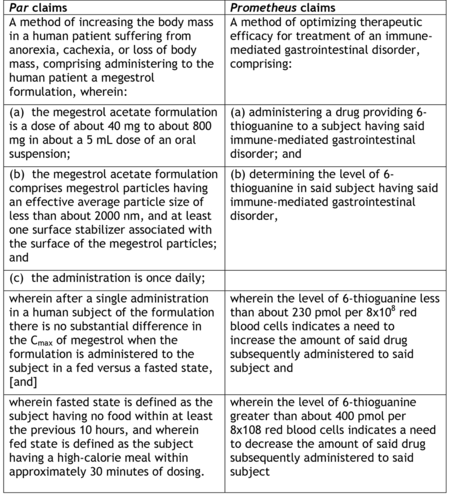
Not to belabor the point, but megestrol acetate formulations and methods for administering them were well established in the art, like the administration step in the Prometheus claims, and formulating megestrol into nanosized particles can easily be encompassed within the Mayo court's "well-established, routine and conventional" rubric. Most telling, the novelty of the Par claims, like the novelty of Prometheus's claims, reside in the "wherein" clauses, which were in Mayo and are likely to be here insufficient to provide that ineluctable "something more" required by the Mayo opinion to provide subject matter eligibility.
Rendering Par's prevailing in its appeal a likely Pyrrhic victory at best and suggesting that the exercise before the Federal Circuit was merely a way station to a negotiated settlement. No doubt such a settlement will then be subject to FTC scrutiny under FTC v. Actavis to determine whether settling raises antitrust liability.
Par Pharmaceutical, Inc. v. TWi Pharmaceuticals, Inc. (Fed. Cir. 2014)
Panel: Circuit Judges O'Malley, Wallach, and Hughes
Opinion by Circuit Judge O'Malley

Leave a comment