Asserts
Affirmative Defenses and Antitrust Counterclaims and Asks for Declaratory
Judgment
By
Kevin E. Noonan —
 On
On
Monday Ambry filed its Answer to Myriad's complaint for patent infringement, and
asserted patent misuse as an affirmative defense. Ambry also asked the District Court for a
declaratory judgment of non-infringement and invalidity of all patents Myriad
has asserted, and raised antitrust counterclaims under the Sherman and Clayton
Acts.
Procedurally,
Ambry raised no challenge to jurisdiction or venue, but denied that the named
plaintiffs are the owners or co-owners of the asserted patents, and admitted only
that it provides genetic diagnostic testing services including for its
BRCAplus, Breastnext, CancerNext, and OvaNext tests as of June 13, 2013.
Substantively,
Ambry's Answer denies that any of the patents were duly and legally issued,
that the claims are valid or infringed, or that Myriad has suffered any damages. Ambry contends as affirmative defenses that Myriad
fails to state a claim for relief, and that the patents are invalid under §§ 102,
103, 112, that the asserted claims do not recite patent-eligible subject matter
under § 101, and that Myriad's asserted claims are unenforceable under the
doctrine of patent misuse. The Answer
asserts no factual or legal grounds for any of its affirmative defenses.
It
is in Ambry's counterclaims that things get interesting. Ambry asserts that it is "a CAP-accredited,
CLIA-certified commercial clinical laboratory, a recognized leader in diagnostic
and contract genomic services specializing in the application of new
technologies to molecular diagnostics and genetics research." Its counterclaims are based on Sections 4 and
16 of the Clayton Act, 15 U.S.C. §§ 15 and 26, and seek treble damages and
injunctive relief under the Sherman Act, 15 U.S.C. § 2. Ambry also seeks a declaratory judgment that
all claims of the patents asserted by Myriad are invalid and not infringed,
either directly or through inducement (presumably, under 35 U.S.C. § 271(b)). There are no allegations of unenforceability
for inequitable conduct.
In
its background facts, Ambry asserts the following allegations:
• Myriad is maintaining its monopoly in violation of the antitrust laws through
"bad faith" enforcement of its patents
These
assertions are based on Ambry's allegations that Myriad "knows" its
patents are invalid under the Supreme Court's AMP v. Myriad and Mayo v.
Prometheus decisions and the Federal Circuit's Myriad decision regarding method claims not asserted in this
litigation. Ambry characterizes this
litigation as a continuation of "sharp and overreaching practices to
wrongfully monopolize the diagnostic testing of human BRCA1 and BRCA2
genes in the United States and to attempt to injure any competitor who dares
to challenge Myriad's monopoly." These "sharp" practices include
"(1) using research funded by public money to file patents over alleged
inventions that the Supreme Court and the Federal Circuit have confirmed never
should have been patented [an ex post
facto argument that ignores the University of Utah's obligations to file
such patents under the Bayh-Dole Act], (2) using those patents to intimidate
and chill competition in the BRCA1 and BRCA2 genetic screening
markets in the late 1990s to ensure monopoly profits, (3) taking patients'
personal BRCA1 and BRCA2 genetic sequence data and depriving the
public of access to that data to inhibit competition, and (4) using sales and
marketing tactics with genetic counselors and payors to intentionally misrepresent
the accuracy and reimbursement of Ambry's BRCA1 and BRCA2 diagnostic
tests." In Ambry's telling of the
tale, the Utah inventors benefited from international efforts to identify
breast cancer-specific genes, including identification of chromosome 17 as the
site of BRCA1 by Mary Claire King. This,
in Ambry's telling, was the critical observation, after which "standard
and well-known techniques could be used to sequence the gene." This
reading minimizes the vast difference between identifying a chromosome, or even
a chromosomal region, genetically linked to a disease or disorder and isolating
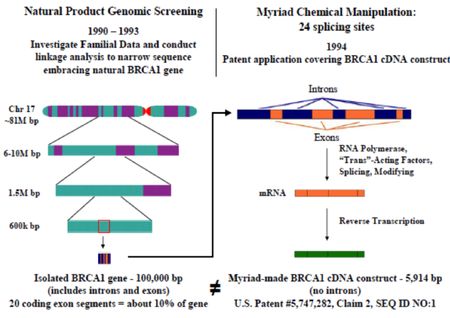
and sequencing the gene, as illustrated below (graphic courtesy of Courtenay Brinckerhoff):
The
situation with the BRCA2 gene was similar, according to Ambry, with the
Stratton group identifying chromosome 13 as the locus of that gene, and the
collaborative nature of work towards its identification. In addition, Ambry asserts that the Stratton
group also isolated and sequenced BRCA2, at about the same time that Myriad
announced its isolation of the gene. Ambry
also emphasizes the amount of "public and private" grant monies
received by the Skolnick group ("$5 million in 1992, $8 million in 1993,
and $9 million in 1994"), and later in their complaint alleges that Myriad
attempted to deprive NIH researchers credit for inventorship (later rectified)
and royalties (allegedly still owed these inventors).
Following
these events, Ambry asserts that Myriad "quickly set out to commercialize
its discoveries," inter alia, by applying for patents on the isolated
BRCA1 and BCRA2 genes. According to
Ambry:
These patents rest on patenting the isolated DNA sequence of the
BRCA1 and BRCA2 genes. The claims at issue in this case do not
cover novel diagnostic tools or methods used in genetic testing. Nor are they analogous
to patents on novel medical instruments. Rather, these claims attempt to confer
upon Myriad the exclusive right to read human BRCA1 and BRCA2 sequences.
(As in the ALCU's lawsuit, Ambry reduces the actual methods used to determine a
genetic sequence to merely "reading" a sequence.) Departing from factual allegations, Ambry
proceeds to argue that Myriad's claims "either attempt to assert broad
ownership over all human DNA sequences that can be used to amplify and sequence
BRCA1 and BRCA2, or merely append routine steps to the patent
claims, which would necessarily be conducted while assessing the biological
relationships between mutations in the BRCA1 and or BRCA2 genes
and the predisposition to cancer." Further, Myriad's patenting strategy "is an overt attempt to
convert these natural biological phenomena into patentable inventions using the
patent language of a 'method' or 'process.'" "In other words,
the patent claims are directed to routine biological procedures and
correlations generically such that it is effectively impossible for anyone in
the United States in any meaningful way to 'make' or 'use'
natural forms of isolated DNA molecules encoding the BRCA1 and BRCA2 genes
in humans without infringing Myriad's patents," features that, in Ambry's
view should apply to any genetic diagnostic method and, indeed, almost all
diagnostic method claims.
Returning
to its characterization of the facts, Ambry asserts that Myriad "set up
$30 million diagnostics laboratory" and provided three tests: a comprehensive
assessment of both BRCA1 and BRCA 2 (costing $2400); a "single site"
test for mutations "previously identified in another family member" ($395);
and a "multisite" test specific for members of the Ashkenazi Jewish
population. Ambry also sets out Myriad's
efforts to assemble "a group of laboratories, health insurers, sales teams
and doctors to market and sell its tests," resulting in $20 million in
revenue in the first three quarters of 1997 alone. At the same time, according to Ambry, Myriad
was threatening competitors (including those at the Genetic Diagnostic Testing
Laboratory at the University of Pennsylvania) with infringement lawsuits for
offering BRCA gene testing, and filed such a lawsuit against the University,
which settled with an agreement that university laboratories would not offer
these tests. Interestingly, Ambry's
allegations of Myriad's malfeasance include a settlement of litigation between
Myriad and OncorMed that resulted in Myriad acquiring OncorMed's patents "to
extend its monopoly."
• Myriad eschews considerations of affordability, test accuracy and
recommendations for genetic testing
These
allegations are based on the following facts according to Ambry. Compared with OncorMed, Myriad offered a
comprehensive rather than a "tiered" approach to testing, thus
requiring the $2400 test rather than tests of fewer mutations offered by
OncorMed costing around $500. (Of
course, when testing a "naïve" patient, i.e., one whose family
history with regard to the existence of BCRA gene mutations was unknown, Myriad's
testing would provide the best information on whether the patient was at risk
for inherited breast cancer.) Ambry also
notes that while OncorMed complied with recommendations of a Federal Task
Force on Genetic Testing (issued March 1996, at the dawn of the generic testing
era), Myriad did (and does) not. (A
similar situation exists for recommendation from the American Society of
Clinical Oncologists, issued May, 1996.) Also in this section of the
complaint are allegations that Myriad "misses" "up to 12%"
of certain types of genetic variations (specifically, rearrangements, deletions
and insertions), citing Congressional testimony from interested parties
(including Wendy Chung, one of the ALCU plaintiffs) attributing delays in this
testing to Myriad's "monopoly."
• There exists significant opposition to Myriad's business model of patenting
gene sequences and establishing a private database
This
portion of Ambry's counterclaims highlights Myriad's decision in 2004 not to
contribute additional information on BRCA gene mutations obtained through is
testing to a public repository of breast cancer-specific mutations, and the
reaction of "[p]oliticians, clinicians, breast cancer advocacy groups and
commentators" who oppose this decision (including the "Sharing
Clinical Reports Project"). Also mentioned is the ACLU's lawsuit and the
Supreme Court's decision invalidating five of nine claims at issue before the
Court.
• Myriad is aware that its asserted claims on BCRA1 and BCRA2 genes are invalid
under recent Supreme Court and Federal Circuit cases
In
this portion of the "factual" background for its counterclaims, Ambry
recites the history and outcomes of the ACLU's Myriad litigation, from the District Court through the Supreme
Court's June 13th decision, as well as the effects of the Court's Mayo decision on this lawsuit. (The history of these lawsuits have been
extensively discussed and Ambry's characterization thereof will not be set
forth here.) This history is recited
here as evidence that "rebuts and presumption [that] Myriad [] brought
this suit in good faith."
As
a consequence of these litigations, Ambry contends that "[Myriad's]
asserted composition claims are not patent eligible, because they are identical
to sequences that occur in nature (a variation of the Department of Justice's "magic
microscope" test not explicitly relied upon by the Court). According to Ambry, Myriad's asserted claims
reciting BRCA gene-specific primers "on their faces fail to recite
patentable subject matter" because the Supreme Court in its Myriad decision "unequivocally
excluded from patentable subject matter synthetic DNA 'that may be
indistinguishable from natural DNA.'" (This is probably Ambry's strongest argument.) Merely being "synthetic" is not
enough to render these primers patent eligible, according to Ambry: "even
though the probe or the primer may have been synthesized in a laboratory, [it
is not patent eligible] so long as it mirrors, in whole or in part, the genomic
DNA of the BRCA1 or BRCA2 genes."
Turning
to Myriad's asserted method claims, Ambry's argument is on less firm
ground. According to Ambry, these claims
are also invalid under Myriad (and Mayo) because they recite "two
biochemical processes [that] were well known in the art." However, what was "routine,
well-understood and conventional" in Mayo was not just the type of methods applied in those claims
but the actual application of the recited methods themselves (i.e., testing
blood from patients administered 6-thioguanine to determine drug or metabolite
levels). Here, in contrast, Ambry's
argument is that these "two biochemical processes" (amplifying and
sequencing DNA) were generically
known, which it true. But the
application of these methods to the BCRA genes was not "routine, well-understood
and conventional" prior to Myriad's patent filing because the subject
matter, the BRCA genes, were unknown in the art. Ambry attempts to overcome this logical
deficiency by equating the BRCA genes to be a "law of nature"; while
the association of specific mutations with breast cancer may be such a law, the
genes themselves are not. In keeping
with the doctrinal laxness of the Court's Mayo decision, Ambry's arguments in
this regard include not only subject matter ineligibility but the purported
lack of novelty and obviousness of Myriad's methods, ignoring the fact that
none of these deficiencies applies to the knowledge of the BCRA genes'
existence in the prior art. Indeed, it
is much more consistent with the Supreme Court's Myriad decision that such
claims are patent-eligible, in view
of the Court's statement that:
Similarly, this case does not involve patents on new applications of knowledge
about the BRCA1 and BRCA2 genes. Judge Bryson aptly noted that, "[a]s the
first party with knowledge of the [BRCA1 and BRCA2] sequences, Myriad was in an
excellent position to claim applications of that knowledge. Many of its
unchallenged claims are limited to such applications."
Regardless,
Ambry contends that Myriad filed its lawsuit "to enforce its invalid patents
in bad faith in order to keep competitors from entering the market,"
citing Myriad's intention to maintain the "status quo" (which,
according to Ambry is Myriad's "wrongful monopoly") as set forth in
its brief in support of its preliminary injunction motion. Ambry also asserts as an improper motive for
the lawsuit its intention to contribute the results of its BRCA gene testing to
public databases. In Ambry's view,
Myriad's failure to do the same constitutes an attempt to use "its invalid
patents to maintain as secret patients' gene sequences (which do not belong to
Myriad) in an attempt to limit competition." And Ambry also asserts that Myriad is using
the lawsuit to delay market entry by competitors other than Ambry, and to
incorporate aspects of its own technology into Myriad's testing, resulting in
an expropriation of "Ambry's superior screening services, which utilize
more sensitive, efficient and cost-effective next-generation sequencing
technologies."
Finally
in this portion of Ambry's counterclaims is the allegation that Myriad has "market
power" and that its activities have harmed the market. This portion of Ambry's counterclaims assert
that the market is diagnostic genetic testing for BCRA gene mutations in the
United States, for which "[t]here are no products that are reasonably
interchangeable substitutes for [BRCA] genetic tests." BRCA testing is also subject to high barriers
to entry, including "the various patents Myriad holds, the technological
know-how for designing and running genetic tests to identify mutations in BRCA1
and BRCA2, and establishing relationships with genetic counselors
and hospitals, many of which are already heavily utilizing Myriad's tests." Myriad has an "over 90% market share"
for BRCA genetic testing and "possesses the power to control prices and
exclude competitors." And the
public suffers because Myriad's tests are "nearly 100% higher" in
costs than Ambry's. Additionally, Ambry
contends that Myriad's contention that Ambry's testing has "variant of
unknown significance (VUS)" rates of 10-30% are false, and that Myriad
knowing this falsity has had its employees conduct a campaign of informing
genetic counselors of this false VUS rate for Ambry's BRCA testing. And these allegations (which are used to
support Ambry's antitrust counterclaims) extend past Myriad: "Plaintiffs
in this action are aware of the anticompetitive and exclusionary conduct by
Myriad [and] that each and every Plaintiff is aware that the claims of the
asserted patents are facially invalid [and] that each and every plaintiff was
involved in the decision to bring this lawsuit, notwithstanding that the
asserted claims are facially invalid."
Count
1 of Ambry's counterclaims is brought under Section 2 of the Sherman Antitrust
Act (against Myriad only). According to
this counterclaim:
126. Myriad is willfully maintaining its monopoly through exclusionary conduct
as distinguished from growth or development as a consequence of a superior
product, business acumen, or historic accident. Myriad seeks to maintain its
monopoly position through various anticompetitive conducts described above,
including through the bad faith enforcement of its facially invalid patents.
127. Myriad was aware before filing its Complaint in this action that the claims it
was asserting are invalid per Myriad and Prometheus.
128. Through its exclusionary and anticompetitive conduct, Myriad has acquired and
maintained its monopoly in the relevant market for genetic testing for patients
seeking analysis of their full BRCA1 and BRCA2 gene sequences. Myriad has operated in this manner free from competition because of the high
barriers to entry that exist in the market, including Myriad's invalid patents,
the technological know-how to design and run genetic tests, and the actions of
Myriad's employees.
Ambry
alleges it has suffered "substantial injury to its business and property"
as a consequence, and that "[c]ompetition in the relevant market" has
similarly suffered "[d]ue to Myriad's anticompetitive conduct."
Count
2 is brought under the Sherman Act for "attempted" monopolization for
substantially the same market behavior, Ambry alleging that Myriad acted "with
a specific intent to monopolize the relevant market through the assertion of
facially invalid patent claims and the anticompetitive conduct of its
employees."
Ambry does not expressly assert that the patents
were obtained through fraud, or that the litigation is brought as a sham, but
rather that the effects of Myriad's assertion of patent claims Ambry asserts
are invalid under recent Federal Circuit or Supreme Court precedent should
incur antitrust liability. In this
regard no better authority that Chief Justice Roberts may be consulted
regarding the relevant legal standard:
The point of patent law is to grant
limited monopolies as a way of encouraging innovation. Thus, a patent grants "the
right to exclude others from profiting by the patented invention." Dawson Chemical
Co. v. Rohm & Haas Co., 448 U. S. 176, 215 (1980). In doing so it provides
an exception to antitrust law, and the scope of the patent — i.e., the rights
conferred by the patent — forms the zone within which the patent holder may
operate without facing antitrust liability.
This should go without saying, in part
because we've said it so many times. Walker Process Equipment, Inc. v. Food
Machinery & Chemical Corp., 382 U. S. 172, 177 (1965) ("'A patent . .
. is an exception to the general rule against monopolies'"); United States
v. Line Material Co., 333 U. S. 287, 300 (1948) ("[T]he precise terms of
the grant define the limits of a patentee's monopoly and the area in which the
patentee is freed from competition"); United States v. General Elec. Co.,
272 U. S. 476, 485 (1926) ("It is only when . . . [the patentee] steps out
of the scope of his patent rights" that he comes within the operation of
the Sherman Act); Simpson v. Union Oil Co. of Cal., 377 U. S. 13, 24 (1964)
(similar).
The key, of course, is that the patent
holder — when doing anything, including settling — must act within the scope of the
patent. If its actions go beyond the monopoly powers conferred by the patent,
we have held that such actions are subject to antitrust scrutiny. See, e.g., United
States v. Singer Mfg. Co., 374 U. S. 174, 196–197 (1963). If its actions are
within the scope of the patent, they are not subject to antitrust scrutiny,
with two exceptions concededly not applicable here: (1) when the parties settle
sham litigation, cf. Professional Real Estate Investors, Inc. v. Columbia
Pictures Industries, Inc., 508 U. S. 49, 60–61 (1993); and (2) when the
litigation involves a patent obtained through fraud on the Patent and Trademark
Office. Walker Process Equipment, supra, at 177.
FTC
v. Activis (Roberts, CJ, dissenting).
The other asserted counterclaims comprise asking
the Court for a declaratory judgment of non-infringement and invalidity of each
of the asserted patents.
Ambry asks for a jury trial, declaratory judgment,
an injunction under Section 16 of the Clayton Act to prevent Myriad or the
other plaintiffs from enforcing the patents-in-suit, treble damages on Ambry's
Sherman Act antitrust counterclaims, and costs and attorneys' fees.

Leave a comment