By Kevin E. Noonan —
 The old adage "Bad cases make bad law" is invoked when the facts of a case lead a court to rule in favor of the particular entities before it rather than applying the law consistently. (Although anyone familiar with recent Supreme Court patent jurisprudence would perhaps not be amiss in thinking the adage should be adapted to "Bad analogies make bad law.") The current Court (or at least a 5-member majority) showed on Monday that it could be immune to these equities in reversing a $21 million judgment to a New Hampshire woman disfigured as a consequence of taking a generic formulation of sulindac, a non-steroidal anti-inflammatory drug (or NSAID). Once again, the Court waded into the waters of drug regulatory law and the interplay between the provisions of the Federal Food, Drug, and Cosmetic Act (FDCA) and Hatch-Waxman Act and state, common law products liability sounding in tort, with the Court deciding that state law was preempted (to the detriment of the patient's recovery for the "tragic circumstances" of her disfigurement).
The old adage "Bad cases make bad law" is invoked when the facts of a case lead a court to rule in favor of the particular entities before it rather than applying the law consistently. (Although anyone familiar with recent Supreme Court patent jurisprudence would perhaps not be amiss in thinking the adage should be adapted to "Bad analogies make bad law.") The current Court (or at least a 5-member majority) showed on Monday that it could be immune to these equities in reversing a $21 million judgment to a New Hampshire woman disfigured as a consequence of taking a generic formulation of sulindac, a non-steroidal anti-inflammatory drug (or NSAID). Once again, the Court waded into the waters of drug regulatory law and the interplay between the provisions of the Federal Food, Drug, and Cosmetic Act (FDCA) and Hatch-Waxman Act and state, common law products liability sounding in tort, with the Court deciding that state law was preempted (to the detriment of the patient's recovery for the "tragic circumstances" of her disfigurement).
The case arose from administration of a generic version of Clinoril (the branded version of the drug sulindac):
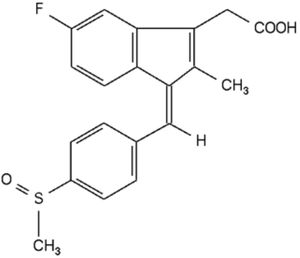
made by Mutual Pharmaceutical to Respondent Karen L. Bartlett. Ms. Bartlett suffered a known — albeit rare — side effect of sulindac administration (as well as other NSAIDs), toxic epidermal necrolysis, a skin hypersensitivity reaction. The consequences for Ms. Bartlett were "horrific" according to the Court: "[s]ixty to sixty-five percent of the surface of respondent's body deteriorated, was burned off, or turned into an open wound. She spent months in a medically induced coma, underwent 12 eye surgeries, and was tube-fed for a year. She is now severely disfigured, has a number of physical disabilities, and is nearly blind." Significant for the Court's decision, at the time she was administered the drug, the drug label did not contain any specific warning regarding this type of side effect (although the FDA required the package insert to contain a warning regarding the risk of developing this condition); the Agency later required this warning to be on the drug label.
Ms. Bartlett sued in an action eventually transferred to Federal court under state law claims characterized by the Court as "both failure-to-warn and design-defect claims," but the District Court dismissed the former cause of action following her physician's admission that he had not read either the label or package insert. Ms. Bartlett prevailed on her design defect claim, which under New Hampshire law "requires manufacturers to ensure that the products they design, manufacture, and sell are not 'unreasonably dangerous.'" According to the Court:
New Hampshire imposes design-defect liability only where "the design of the product created a defective condition unreasonably dangerous to the user," citing Vautour v. Body Masters Sports Industries, Inc., 147 N. H. 150, 153, 784 A. 2d 1178, 1181 (2001); Chellman v. Saab-Scania AB, 138 N. H. 73, 7, 637 A. 2d 148, 150 (1993). To determine whether a product is "unreasonably dangerous," the New Hampshire Supreme Court employs a "risk-utility approach" under which "a product is defective as designed if the magnitude of the danger outweighs the utility of the product." Vautour, supra, at 154, 784 A. 2d, at 1182 (internal quotation marks omitted). That risk-utility approach requires a "multifaceted balancing process involving evaluation of many conflicting factors." Ibid. (internal quotation marks omitted); see also Thibault, supra, at 809, 395 A. 2d, at 847 (same).
While the set of factors to be considered is ultimately an open one, the New Hampshire Supreme Court has repeatedly identified three factors as germane to the risk-utility inquiry: "the usefulness and desirability of the product to the public as a whole, whether the risk of danger could have been reduced without significantly affecting either the product's effectiveness or manufacturing cost, and the presence and efficacy of a warning to avoid an unreasonable risk of harm from hidden dangers or from foreseeable uses." Vautour, supra, at 154, 784 A. 2d, at 1182; see also Price, supra, at 389, 702 A. 2d, at 333 (same); Chellman, supra, at 77-78, 637 A. 2d, at 150 (same).
Moreover, according to the Court this form of liability, while being "strict" product liability is not "absolute" liability; the distinction is that the former imposes a "duty of care" on manufacturers as opposed to the latter form of liability, which is a way states can "spread the risk" of injury due to design or manufacturing defects. This distinction is relevant, because the Court's determination that Mutual was under a common law-defined duty in New Hampshire implicated the three ways the company could comply with state law, and how two of the three were impossible under Federal law.
 The Court's opinion, by Justice Alito joined by Chief Justice Roberts and Justices Scalia, Kennedy, and Thomas, found that Mutual could comply with its state-law imposed duty either by making chemical changes to the drug or changing the drug label (here, by including the more stringent warnings from the package insert on the label, as the FDA was later to do). The majority found that changing sulindac chemically was impossible both for reasons of "basic chemistry" (i.e., there is little that could be changed in the molecule without changing it into a different molecule) and FDA regulatory law, which required the generic compound to be a copy of the branded drug and to have certain pharmacological properties in common with the branded embodiment. 21 U. S. C. §§ 355(j)(2)(A)(ii), (iii) and (iv), 21 U. S. C. §§ 355(j)(8). The impossibility of changing the label is a direct consequence of the FDA requirement that a generic drug have the same label as the branded version. § 355(j)(2)(A)(v). And a generic drug manufacturer is barred from unilaterally changing either the drug itself, its formulation or its label. 21 CFR § 314.70(b)(2)(i). Thus, in this case there was a direct conflict between what was required to comply with the state-imposed duties under its tort law and the requirements of Federal drug regulatory law.
The Court's opinion, by Justice Alito joined by Chief Justice Roberts and Justices Scalia, Kennedy, and Thomas, found that Mutual could comply with its state-law imposed duty either by making chemical changes to the drug or changing the drug label (here, by including the more stringent warnings from the package insert on the label, as the FDA was later to do). The majority found that changing sulindac chemically was impossible both for reasons of "basic chemistry" (i.e., there is little that could be changed in the molecule without changing it into a different molecule) and FDA regulatory law, which required the generic compound to be a copy of the branded drug and to have certain pharmacological properties in common with the branded embodiment. 21 U. S. C. §§ 355(j)(2)(A)(ii), (iii) and (iv), 21 U. S. C. §§ 355(j)(8). The impossibility of changing the label is a direct consequence of the FDA requirement that a generic drug have the same label as the branded version. § 355(j)(2)(A)(v). And a generic drug manufacturer is barred from unilaterally changing either the drug itself, its formulation or its label. 21 CFR § 314.70(b)(2)(i). Thus, in this case there was a direct conflict between what was required to comply with the state-imposed duties under its tort law and the requirements of Federal drug regulatory law.
Under these circumstances, the Court held that Federal law prevails and preempts state tort liability, citing Maryland v. Louisiana, 451 U. S. 725, 746 (1981), for the principle that "[u]nder Supremacy Clause, state laws that require a private party to violate federal law are pre-empted and, thus, are 'without effect.'" And the Court rejected the third possibility open to Mutual, which was to stop selling generic sulindac in New Hampshire, saying that if this were the law "impossibility preemption" would be "all but meaningless," citing its recent opinion in PLIVA, Inc. v. Mensing, 564 U. S. ___ (2011), a case involving state law liability on "failure to warn" grounds. The Court expressly rejected this alternative in the PLIVA case, saying that "[a]dopting the [appellate court's] stop-selling rationale would mean that not only PLIVA, but also the vast majority — if not all — of the cases in which the Court has found impossibility pre-emption, were wrongly decided."
That, of course, would never do. As a consequence, Ms. Bartlett is without a remedy against the generic drug manufacturer, an outcome the majority regrets but does not use to come to a different conclusion:
This case arises out of tragic circumstances. A combination of factors combined to produce the rare and devastating injuries that respondent suffered: the FDA's decision to approve the sale of sulindac and the warnings that accompanied the drug at the time it was prescribed, the decision by respondent's physician to prescribe sulindac despite its known risks, and Congress' decision to regulate the manufacture and sale of generic drugs in a way that reduces their cost to patients but leaves generic drug manufacturers incapable of modifying either the drugs' compositions or their warnings. Respondent's situation is tragic and evokes deep sympathy, but a straightforward application of pre-emption law requires that the judgment below be reversed.
Four Justices dissented. Justice Breyer, joined by Justice Kagan, writes that there is a fourth alternative, that is for a company to shoulder the burden of "paying a sizable damages" award as a remedy in instances where a rare side effect causes harm to a patient. This opinion is informed, in part, by an amicus brief submitted by two former FDA Commissioners, who inform the Court that "the FDA has long believed that state tort litigation can 'supplemen[t] the agency's regulatory and enforcement activities.'" Justice Sotomayor, joined by Justice Ginsburg, is more vociferous, accusing the majority of unnecessarily crafting a rule that "immunize[d] generic drug manufacturers from state-law failure-to-warn claims" in PLIVA v. Mensing and, here, "unnecessarily and unwisely extend[ing] its holding in Mensing to pre-empt New Hampshire's law governing design-defects with respect to generic drugs." "Of greater consequence," the Justice writes:
[T]he Court appears to justify its revision of respondent Karen Bartlett's state-law claim through an implicit and undefended assumption that federal law gives pharmaceutical companies a right to sell a federally approved drug free from common-law liability. Remarkably, the Court derives this proposition from a federal law that, in order to protect consumers, prohibits manufacturers from distributing new drugs in commerce without federal regulatory approval, and specifically disavows any intent to displace state law absent a direct and positive conflict.
The majority has gone astray, according to the dissent, for failing to apply the "two cornerstones of our pre-emption jurisprudence": to discern the "purpose of Congress" in enacting the statute, and to require a "clear and manifest" Congressional intention to supersede the "historic police powers of the States" as embodied in the type of common law tort liability at issue in this case. The Justice sees this decision as being in contrast to the Court's (correct) decision in Wyeth v. Levine, where state tort law liability was not preempted by Federal drug regulatory law. State tort law is a complement to Federal drug law in protecting consumers, a situation Congress has not seen fit to alter or correct:
Congress' preservation of a role for state law generally, and common-law remedies specifically, reflects a realistic understanding of the limitations of ex ante federal regulatory review in this context. On its own, even rigorous preapproval clinical testing of drugs is "generally . . . incapable of detecting adverse effects that occur infrequently, have long latency periods, or affect subpopulations not included or adequately represented in the studies," citing Kessler & Vladeck, A Critical Examination of the FDA's Efforts to Preempt Failure-to-Warn Claims, 96 Geo. L. J. 461, 471 (2008); see National Academies, Institute of Medicine, The Future of Drug Safety: Promoting and Protecting the Health of the Public 37-38 (2007).
Her dissent also notes that "the FDA, which is tasked with monitoring thousands of drugs on the market and considering new drug applications, faces significant resource constraints that limit its ability to protect the public from dangerous drugs," citing Levine, 555 U. S., at 578-579, and n. 11, and that "[t]ort suits can help fill the gaps in federal regulation by 'serv[ing] as a catalyst' to identify previously unknown drug dangers," citing Bates v. Dow Agrosciences LLC, 544 U. S. 431, 451 (2005).
Under these circumstances this dissent finds the facts of this case to be far from the "strict standard for impossibility" that should be required for Mutual to avoid state law tort liability. And as in Justice Breyer's dissent, Justice Sotomayor did not see Mutual's alteratives to be limited to those "impossible" under Federal law:
A manufacturer of a drug that is unreasonably dangerous under New Hampshire law has multiple options: It can change the drug's design or label in an effort to alter its risk-benefit profile, remove the drug from the market, or pay compensation as a cost of doing business. If federal law or the drug's chemical properties take the redesign option off the table, then that does not mean the manufacturer suddenly has a legal obligation under state law to improve the drug's label. Indeed, such a view of state law makes very little sense here because even if Mutual had strengthened its label to fully account for sulindac's risks, the company might still have faced liability for having a defective design. . . . When a manufacturer cannot change the label or when doing so would not make the drug safe, the manufacturer may still choose between exiting the market or continuing to sell while knowing it may have to pay compensation to consumers injured by its product.
While these alternatives may be "unwelcome" from a manufacturer's prospective, "it is a choice that a sovereign State may impose to protect its citizens from dangerous drugs or at least ensure that seriously injured consumers receive compensation."
Ironically, the majority, usually so quick to rely upon Federalism (or "states' rights") principles in other contexts finds state tort law liability to be preempted while the traditionally "liberal" members of the Court would hew to basic Federalism principles in permitting states to protect their citizens under common law tort law. While the majority "saves for another day the question whether a true absolute-liability state-law system could give rise to impossibility pre-emption," it seems that states adopting an "absolute liability" regime that imposes no affirmative duties contrary to Federal law but merely "spreads the risk" might be one way for the states to regain their traditional right to employ the sovereign police powers to impose tort liability that would escape the wide latitude of preemption embodied in the Court majority's decision.
Mutual Pharmaceutical Co. v. Bartlett (2013)
Opinion by Justice Alito, joined by Chief Justice Roberts and Justices Scalia, Kennedy, and Thomas; dissenting opinion by Justice Breyer, joined by Justice Kagan; dissenting opinion by Justice Sotomayor, joined by Justice Ginsburg

Leave a comment