By James DeGiulio —
 With very little downside
With very little downside
and huge upside, exclusivity is the driving force to the huge increase in
first-to-file Paragraph IV filings. Adam Greene and D. Dewey Steadman, both at RBC Capital Markets Corp., conducted
a recent study where they analyzed over 370 court rulings since the beginning
of 2000 to establish the "success rates" of generics (as defined by
the authors). Using PACER, company reports, and RBC Capital Markets estimates
as their sources, the authors looked at company, district court, and judge, as
well as other relevant trends in the industry such as at-risk launches,
authorized generics, and the number of patent settlements. The authors
hypothesize that, based on the incentives of the Hatch-Waxman first-to-file
structure, every patented product would be challenged. As expected, the authors
saw an increase in first-to-file lawsuits in each year since 2003, reaching a
record high of 65 in 2009 (see chart below).
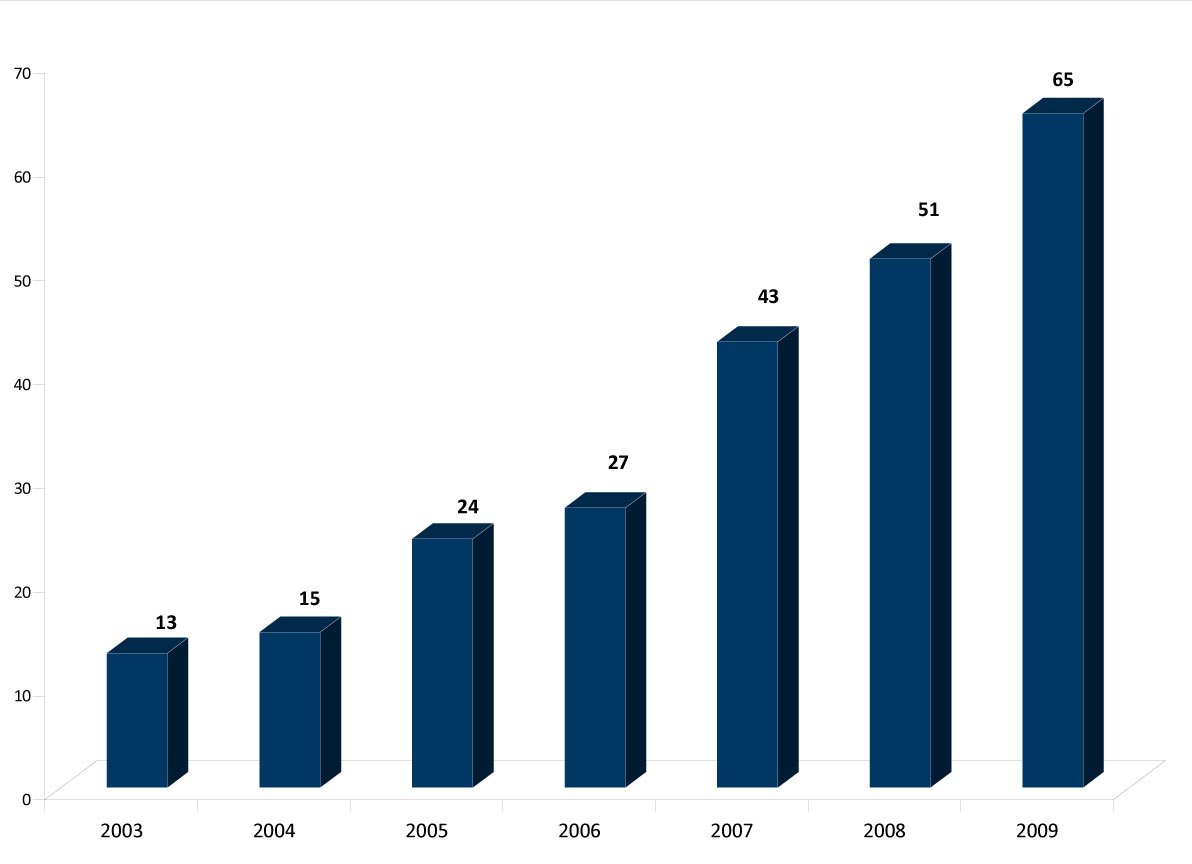 (Exhibit 2: First-to-File Lawsuits, Greene and Steadman, "Pharmaceuticals: Analyzing Litigation Success Rates," January 15, 2010)
(Exhibit 2: First-to-File Lawsuits, Greene and Steadman, "Pharmaceuticals: Analyzing Litigation Success Rates," January 15, 2010)
Perhaps the most
intriguing finding of the study, the authors determined the "success rate"
of generics for Paragraph IV challenges was a healthy 76% (282/370) (see chart below). Importantly, the authors consider a settlement as a "success" for the
generic, as it eliminates uncertainty and expensive legal costs. Dropped cases
were also considered a successful outcome for generics, though the reasoning
for this is not provided in the study. Thus, as long as settlements and dropped
cases are considered a "success" for generics, the study supports the
authors' hypothesis that every patent would be challenged by a generic. With a
76% success rate, the potential payoff of a first-to-file Paragraph IV
challenge is worth the risk of litigation. However, the study finds generics
fare much worse at trial. Of the 171 cases resolved at trial, generics won 82
rulings while losing 89 (48%).

(Exhibit 3: Generic Drug Industry: Litigation Success Rate, Greene and Steadman, "Pharmaceuticals: Analyzing Litigation Success Rates," January 15, 2010)
The authors next
analyzed the track records of generics with five or more resolved Paragraph IV challenges
between 2000 and 2009. Perrigo was found
to have a perfect success record, with a single case won and seven settlements,
once again reflecting the author's classification of all settlements as a
successful outcome. Apotex was
found to have the worst success rate at 43%, but was also the least likely to
settle or have a case dropped. All
company data is presented in the table below.
(Exhibit 4: Best Generic Challengers 2000-2009, Greene and Steadman, "Pharmaceuticals: Analyzing Litigation Success Rates," January 15, 2010)
The authors next evaluated which federal districts
and which judges have the best success rates for generics. A few districts have
never found against generics, though these districts have not heard many cases. These include the Central District of California (8/8), the Eastern District of
New York (2/2), Minnesota (2/2), and the Eastern District of Missouri (1/1). However, the most popular districts (New
Jersey, Delaware, and the Southern District of New York),
which heard nearly 70% of all generic challenges, resulted in a generic winning
percentage of only 36% (see table below). This winning percentage is on par with the authors'
analysis of individual judges. For
judges who have issued 5 or more decisions, the winning rate for generics is
37%.
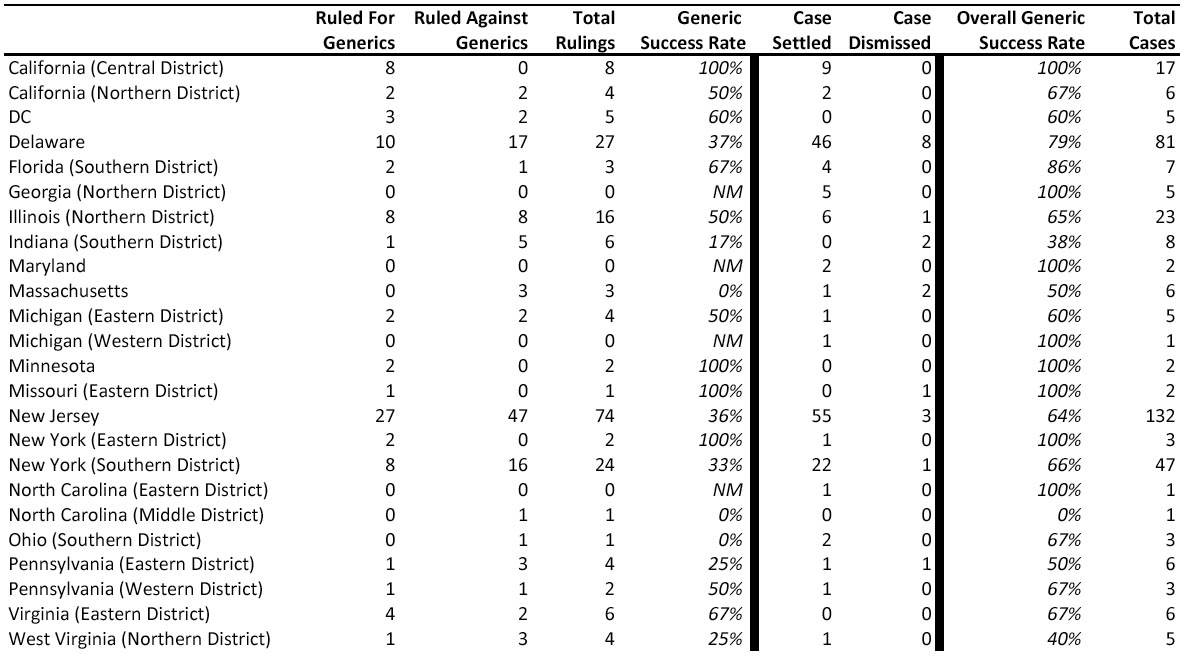
(Exhibit 6: Decisions By Court, Greene and Steadman, "Pharmaceuticals: Analyzing Litigation Success Rates," January 15, 2010)
The authors noticed a small increase in at-risk
launches (defined as any launch without a lower court
ruling) and authorized generics. Further, the authors noticed the number of
non-authorized generic launches doubled from 2008-2009, from 6 to 12, which the
authors hypothesize is due to the increase in settlement agreements a few years
ago.
Finally, the authors
note a trend of increasing settlements from 2003-2009, reaching an all-time
high of 54 in 2009 (see chart below). Teva accounted
for nearly one third of all settlements.
On the brand side, Glaxo and Novartis have settled the most.
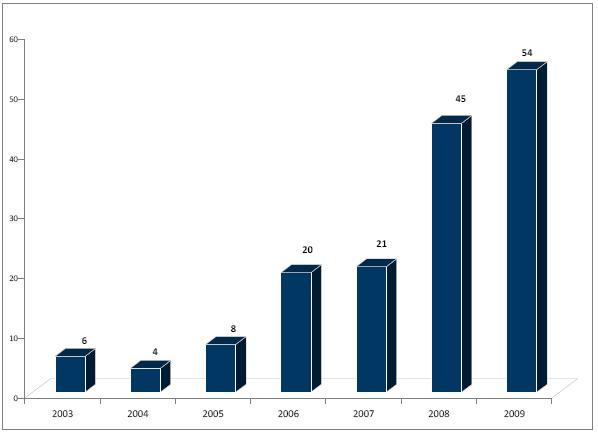
(Exhibit 10: Number of Patent Settlements 2003-2009, Greene and Steadman, "Pharmaceuticals: Analyzing Litigation Success Rates," January 15, 2010)
The authors expect
settlements to remain fairly common, despite the passing of the Kohl Bill by
the Senate Judiciary Committee in October 2009. Their prediction is not surprising, since the authors assume
that settlements are always good for the generic company. From an investment
perspective, this assumption may have merit, since settlements provide clarity
for the company (and shareholders) and avoid the expense and risk of
litigation. However, in practice,
there will certainly be settlements that would not be considered a success for
the generic.
James DeGiulio has a doctorate in molecular biology and genetics from Northwestern University and is a third-year law
student at the Northwestern University School of Law. Dr. DeGiulio
was a member of MBHB's 2009 class of summer associates, and he can be contacted at degiulio@mbhb.com.

Leave a comment