By
Donald Zuhn —
 On
On
Monday, an en banc panel of the
Federal Circuit heard oral argument in Ariad
Pharmaceuticals, Inc. v. Eli Lilly & Co. (see "Ariad v. Lilly:
Oral Argument"). During the hearing, several members of
the Court as well as counsel for the Government focused on perceived
differences between the positions taken by Plaintiffs-Appellees ("Ariad")
in their principal and reply briefs.
(For a discussion of Ariad's principal brief, please see "Next Up: Ariad v. Lilly Rehearing En Banc"). Today, we examine Ariad's reply brief.
Ariad
begins the brief by listing the points on which the parties and amici find common ground, stating that:
Lest it be obscured by rhetoric, it is important
to note that there is much common ground amongst the parties and amici. Specifically, they agree that:
1. The specification must provide a written
description of the invention and of the manner and process of making and using
it.
2. The description requirement of § 112
applies to all claims, whether they were originally present in the application
or not.
3. The description requirement of § 112
polices an applicant's ability to add or amend claims during prosecution.
4. A patent may not claim an invention
that is not described in the specification.
5. The description requirement of § 112
prevents claims that extend beyond the inventor's contribution as set forth in
the specification;
claims
of broad applicability must correspond to an invention that is identified in
the specification by a broadly applicable common principle.
Ariad
then summarizes the "actual disagreement" between the parties as
concerning the issue of:
[W]hether § 112, first paragraph requires
a written description of the invention that is separate from enablement.
Specifically, is the entire written description required by § 112 (i.e.,
the description of the invention, and of the manner and process of making and
using it) measured by the statutory duty to describe the invention in "such
full, clear, concise, and exact terms as to enable any person skilled in the
art to which it pertains . . . to make and use the same," as
Plaintiffs-appellants contend? Or, as urged by Lilly, does § 112 impose a written
description requirement independent of the rest of the statute and measured by
the judicially-created "possession" test of Regents of the University of California v. Eli Lilly & Co., 119
F.3d 1559 (Fed. Cir. 1997) and its progeny?
Ariad's brief
next addresses the language of § 112, first paragraph, providing a table
comparing the statutory constructions proposed by the parties:
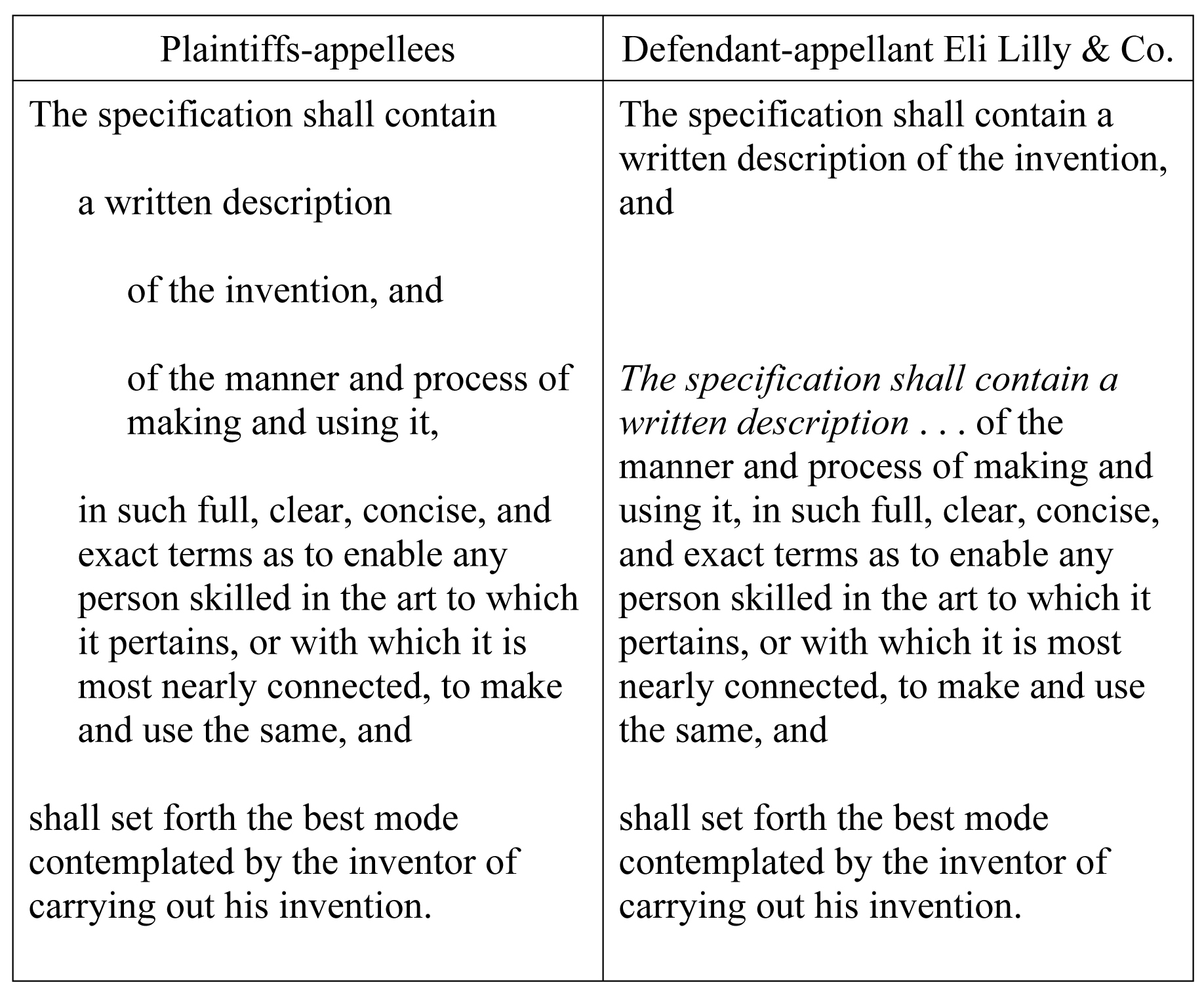
Noting
that "Lilly has effectively rewritten the statute by adding the italicized
words," Ariad contends that Lilly's construction:
[S]evers the "written description
of the invention" not only from the requirement to enable, but also from
the requirement that it be in "full, clear, concise, and exact"
terms. There is no reason for
Congress to demand a written description of "the manner and process of
making and using" the invention that is "full, clear, concise, and
exact" while tolerating a "written description of the invention"
that is incomplete, unclear, long-winded, and inexact.
As
in its principal brief, Ariad examines and compares the statutory language from
the Patent Acts of 1793, 1836, and 1952, and concludes that:
The history of the Patent Act also
shows that, since 1793, the requirement for a description of the invention has
always been linked with the requirement to describe the manner of making and
using the invention, and has been measured by the text that follows, i.e.,
clear, concise and exact, etc.
Ariad's
brief next provides a discussion of relevant Supreme Court precedent. Contending that Lilly "mischaracterizes
the Supreme Court decisions on which it relies," Ariad states that "Supreme
Court precedent both before 1836 and after 1836 provides no support for the
view that the written description requirement is to be separated from its
statutory standard for enablement, let alone that it be judged by this Court's
current 'possession' test."
Ariad notes that Lilly has failed to produce "even one post-1836 Supreme
Court opinion in which the description of an invention claimed or otherwise
identified at the time of filing was found to be deficient under any standard
other than the enablement standard."
According to Ariad, the reason for this failure is that:
The Supreme Court cases have never
decoupled the "written description of the invention" from rest of the
statutory language and have never held that a written description of an
invention sufficient to enable others to practice a claimed invention could
nonetheless fail because the description somehow did not "establish that
the applicant was in possession of the claimed invention."
Ariad
explains that when § 112, first paragraph, is interpreted as proposed in its
briefs, the "description requirement" serves two purposes by: (1) "prevent[ing] patent
applicants from introducing new or amended claims directed to a different
invention than is described in an earlier-filed specification," and (2) "ensur[ing]
that the claims — whether original or amended — match the inventor's
contribution to the art."
With respect to the second objective, Ariad notes that claims
encompassing more than the particular embodiments disclosed in the
specification must be supported by an identification of "the common
principle that defines the invention and distinguishes it from the prior art."
In
the last portion of its reply brief, Ariad outlines how the claims at issue in
the appeal satisfy the "description requirement" of § 112, when §
112, first paragraph, is construed as proposed in Ariad's briefs. Ariad then summarizes its opponent's argument
by stating that:
Under Lilly's view, even though the claims-in-suit
might be directed to novel, useful, non-obvious, and statutory methods, and
even though the '516 patent applicants might have complied with their statutory
duty to provide a written description of their invention "in such full,
clear, concise, and exact terms as to enable any person skilled in the art to
which it pertains . . . to make and use the same," the asserted claims are
nevertheless worthless and void because the '516 patent applicants purportedly
did not comply with a "separate", non-statutory, judicially-devised
duty to describe their invention in terms that a court or jury might deem
sufficient to prove that the '516 patent applicants had "possession"
of the claimed methods to a legally sufficient degree.
Ariad,
however, counters by arguing that:
The discoverer of a new and non-obvious
process, whose use is not tied to particular materials or machines, has never
been restricted in his or her patent protection to the details of particular
instrumentalities that were then the best-known ways of carrying out the
process. Lilly's "written description" arguments are aimed at denying
patent protection to upstream researchers who discover and disclose generally
applicable methods that Lilly would then subsequently exploit.
For additional information regarding this topic, please see:
• "Ariad v. Lilly:
Oral Argument," December 9, 2009
• "Amicus Briefs in Ariad v. Lilly: Regents of University of California et al.," December 6, 2009
• "Amicus Briefs in Ariad v. Lilly: Briefs by Companies," December 6, 2009
• "Amicus Briefs in Ariad v. Lilly: American Intellectual Property Law Association," December 1, 2009
• "Amicus Briefs in Ariad v. Lilly: Federal Circuit Bar Association," November 29, 2009
• "Amicus Briefs in Ariad v. Lilly: Intellectual Property Owners Association," November 25, 2009
• "Amicus Briefs in Ariad v. Lilly: GlaxoSmithKline," November 24, 2009
• "Amicus Briefs in Ariad v. Lilly: United States," November 23, 2009
• "Amicus Briefs in Ariad v. Lilly: Google, Verzion Communications Inc. and Cisco Systems, Inc.," November 22, 2009
• "Amicus Briefs in Ariad v. Lilly: Professor Christopher Holman," November 19, 2009
• "Lilly Files Principal Brief for Ariad v. Lilly Rehearing En Banc," November 16, 2009
• "Next Up: Ariad v. Lilly Rehearing En Banc," November 10, 2009
• "Federal Circuit Grants En Banc Review in Ariad v. Lilly," August 21, 2009
• "Ariad Files Petition for Rhearing in Ariad v. Lilly," June 3, 2009
• "Ariad Decision Voids Attempt to Use Broad Claiming to Avoid the Written Description Requirement," April 14, 2009
• "Ariad Pharmaceuticals, Inc. v. Eli Lilly and Co. (Fed. Cir. 2009)," April 6, 2009

Leave a comment