More Claim Construction Confusion from the Federal
Circuit
By Kevin E. Noonan —
The Federal Circuit demonstrated once again the
quagmire that can exist when lower courts attempt to apply the CAFC's rather
quixotic jurisprudence on claim construction.
 The case, Sanofi-Aventis
The case, Sanofi-Aventis
U.S. LLC v. Sandoz, Inc.(1), arose as the result of ANDA filings by several Defendants for oxaliplatin, the active ingredient in Sanofi's Eloxatin®
colorectal cancer drug. Two
claims of the patent at issue, U.S. Patent No. 5,338,874, were asserted against
the Defendants:
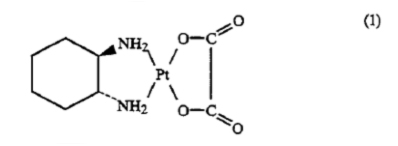
1. Optically-pure
cis-oxalato(trans-l-1,2-cyclohexanediamine) Pt(II) having the general formula
of Formula (1).
(Dependent claim 2 merely added a melting point
limitation and was not further at issue in the case.) The question decided by the Federal Circuit on appeal was
whether the claim term "optically pure" included an affirmative
limitation that "optically pure oxaliplatin" had been "resolved
by means of the HPLC method described" in the specification of the '874
patent.
The District Court had decided that this term did
include the limitation. While
admitting that "[t]here was no
dispute that nothing on the face of the claims of the '874 patent limit[ed] the
claims to 'optically pure oxaliplatin that is produced through the use of HPLC,"
the District Court identified statements in the prosecution history and
disclosure in the specification that supported its interpretation. Specifically, the District Court was
persuaded that Sanofi had distinguished an asserted prior art reference (Kidani
et al.) that purported to disclose "a single isomer [of oxaliplatin]
useful as an antitumor agent" by arguing that the optical purity of the
oxaliplatin in the reference was only 90%. According to the District Court, Sanofi stated during
prosecution that "[o]nly after HPLC resolution (in accordance with the
teachings of the present application) was optical purity obtained." In addition, the District Court pointed
to portions of the specification that compared the optical purity of oxaliplatin
prepared according to prior art methods contained in the asserted reference with
the optical purity of Sanofi's molecule after resolution by HPLC. To the District Court, these
distinctions were sufficient to require that the claim term "optically
pure oxaliplatin" describe only oxaliplatin resolved to optical purity by
HPLC. Accordingly, since the Defendants did not use HPLC to resolve oxaliplatin to optical purity, the District Court granted summary judgment of non-infringement.
 The Federal Circuit disagreed with the lower court's
The Federal Circuit disagreed with the lower court's
claim construction. In a unanimous
(albeit nonprecedential) opinion by Judge Moore, joined by Judges Linn and
Prost, the Court found that the patentee had not disclaimed all embodiments of
optically pure oxaliplatin except those resolved by HPLC. The Court opined as follows:
We have repeatedly warned of "the danger of
reading limitations from the specification into the claim." See, e.g.,
Phillips, 415 F.3d at 1323. "Absent a clear disavowal or contrary
definition in the specification or the prosecution history, the patentee is
entitled to the full scope of its claim language." Home Diagnostics,
Inc. v. Lifescan, Inc., 381 F.3d 1352, 1358 (Fed. Cir. 2004). To narrow the
plain language of a claim, a disclaimer must be clear and unmistakable. Cordis
Corp. v. Boston Scientific Corp., 516 F.3d 1319, 1329 (Fed. Cir. 2009). We
see no such disclaimer in the specification or prosecution history of the '874
patent.
The Court also disagreed that claim 1 of the '874
patent was a product-by-process claim:
The district court relied on Andersen Corp. v.
Fiber Composites, L.L.C., 474 F.3d 1361 (Fed. Cir. 2007), when construing
claim 1 as a product-by-process claim. In Andersen, this court held that
claims to composite structures included a pelletizing process limitation where
the patentee relied on that process both
to define the invention and to distinguish the prior art. . . . We
determined that the specification attributed the claimed physical properties to
the process and that the specification indicated that the pelletizing step was a
requirement, not a preference, of the invention. . . . We further determined
that the patentee had clearly disavowed
other processes during prosecution. . . . [emphasis
added]By contrast, here, the patent specification and
prosecution history focus on the property of the composition (optical purity)
and not the process used to obtain that property. . . . The specification never
asserts that HPLC is required to obtain optically pure oxaliplatin. It
characterizes HPLC as an "illustrative method" and a "representative
process" by which the claimed compound "may be prepared". . . . Moreover,
the specification does not define the property (optical purity) by reference to
the process of purification by HPLC. Thus nothing in the specification limits
the invention to optically pure oxaliplatin purified using HPLC.The prosecution history also illustrates that it is
the optical purity of oxaliplatin that distinguished it from the prior art, not
the process used to obtain that purity. The Examiner rejected the initially
filed claims to oxaliplatin "of optically high purity" as anticipated
or rendered obvious by Kidani.
The Examiner stated that Kidani disclosed "a
single isomer [oxaliplatin] useful as an antitumor agent. Note that since the
single isomer complex was prepared, the optical purity of such material is very
high or almost pure isomer." In response, the applicant . . . explained that it had repeated
Kidani's process "using identical reactant materials and the subsequent
testing thereof. . . . The resultant material was tested and found to be 90%
[oxaliplatin] not optically pure . . . ." Sanofi explained that "[o]nly
after HPLC resolution (in accordance with the teachings of the present
application) was optical purity obtained." Sanofi further asserted that
the products prepared using Kidani's method "do not have the presently
claimed optical purity." Therefore, Sanofi argued that the claimed
oxaliplatin "having high optical purity[] is not found or taught in the
prior art either by inherency or by being obvious thereover." Following a
telephone interview, Sanofi agreed to amend the claims to "optically pure"
oxaliplatin, rather than oxaliplatin "of high optical purity." The
Examiner entered the amendment and allowed the claims, stating that "[t]he
Examiner agrees with applicants that Kidani et al. does not teach[] the
cis-oxalato(trans-l-1,2-cyclohexanediamine)Pt(II) as an optically pure isomer.
It is clear from Kidani et al. that also other isomers can be in the final
product." Thus, Sanofi argued that the
defining feature of the claimed oxaliplatin was its optical purity, not the
HPLC process. Nothing in the prosecution history amounts to a clear and
unmistakable disclaimer of optically pure oxaliplatin prepared using other
(non-HPLC) processes. [emphasis added].
The Federal Circuit vacated the District Court judgment of non-infringement and remanded the
case to the lower court.
The panel's opinion illustrates how difficult it is
for a district court to implement supposedly straightforward rubrics such as
those enunciated by the en banc court
in Phillips v. AWH Corp. The District Court's reasoning was
based on understanding the plain meaning of the term "optically pure,"
a phrase that has no plain meaning outside its specialized meaning in the
chemical arts. The District Court
looked to the specification and the prosecution history to identify how the
patentee defined the term, and in the absence of an express definition used the
examples in the specification and the arguments and amendments made during
prosecution to construe the claim term "optically pure" to require
that optical purity must be achieved using HPLC. The Federal Circuit disagreed and substituted its judgment
for the lower court's, a commonplace occurrence.
The outcome brings to mind the dissenting opinion
of District Court Judge Vaughn
R. Walker, Chief Judge, U.S. District Court for the Northern District of
California, sitting by designation, in Medegen MMS, Inc. v. ICU Medical Inc. (Fed, Cir. 2008) (see "Claim Construction at the Federal Circuit: A District Court Judge's View"). In
that case, Judge Walker asserted that claim construction presented "a
conundrum" to district courts. This conundrum arose from these "twin
axioms" of claim construction: "[o]n the one hand, claims 'must
be read in view of the specification'," citing Markman, while "[o]n
the other hand, it is improper to read a limitation from the specification into
the claims," citing Arlington Indus. Inc. v. Bridgeport Fittings, Inc. The problem with these axioms,
according to Judge Walker, is that "the axioms themselves seldom provide
an answer, but instead merely frame the question to be resolved." While recognizing the sound policy reasons for a patent drafter to be required
to include "all claim limitations in the claims of a patent," he also
admonished that "patents must be read as well as written." Citing Phillips, he asserted that this analysis must be performed with
due consideration of a term's "ordinary and customary meaning"
informed by the "context of the entire patent, including the
specification."
Judge Walker's
dissent raised the essential paradox of the Federal Circuit's practice under Cybor Corp. v. FAS Technologies, Inc. of giving no deference to claim
construction by district courts: "[a]s a jurist more accustomed to
working on the front lines of patent litigation than reviewing decisions from
above, it is my experience that claim construction — determining how one of
ordinary skill in the art would understand the patent at the time of invention
— often requires making fact-like determinations not well suited to appellate
review." The "uniformity and predictability" desired by
the CAFC as a general, guiding principle is not always best served in the
individual determinations necessary to construe claim terms. In this he
cited with approval Judge Lourie's point in Phillips, that while there
is (and perhaps cannot be) formal deference by the CAFC to a district court's
claim construction, it is better policy for the Federal Circuit to affirm "in
the absence of a strong conviction of error."
Current academic
research suggests that Federal Circuit affirmance rates of district court claim
construction has "improved" to from 60-70%. However, each Federal Circuit panel addresses, and resolves,
specific claim construction issues in any particular case in its own way,
introducing the kind of uncertainty that the Federal Circuit was created to curtail. The inconsistencies resulting from the Federal Circuit's "no deference"
standard of review can be expected to continue to produce the type of results
decried by Judge Walker in Medegen and, arguably, that were obtained in Sanofi-Aventis v. Sandoz.
(1) Additional defendants included Teva Parenteral
Medicines, Inc., Teva Pharmaceuticals USA, Inc., and Pharmachemie BV; Mayne
Pharma Ltd., Mayne Pharma (USA) Inc,, Hospira Australia Pty Ltd., and Hospira,
Inc.; W.C. Heraeus GMBH; APP Pharmaceuticals, Inc. and Abraxis Bioscience,
Inc.; Actavis Totowa LLC, Actavis, Inc. and Actavis Group HF; Fresenius Kabi
Oncology PLC and Fresenius Kabi Pharma Ltd.; Sun Pharmaceutical Industries Ltd.
and Caraco Pharmaceutical Laboratories, Ltd.; Ebewe Pharma GES.M.B.H. NFG HG;
and Mustafa Nevzat Ilac Sanayii A.S., Par Pharmaceutical Companies, Inc. and
Par Pharmaceutical, Inc.
Sanofi-Aventis U.S. LLC v. Sandoz, Inc. (Fed. Cir.
2009)
Nonprecedential disposition
Panel: Circuit Judges Linn, Prost, and Moore
Opinion by Circuit Judge Moore

Leave a comment