By Kevin E. Noonan —
 BioWorld Today posted an article last week about
BioWorld Today posted an article last week about
the top 25 biotech drugs currently on the market. Written by Michael Harris, Executive Editor, the piece
represents a précis of a longer report by BioWorld, entitled "Market-Leading
Biotechnology Drugs 2009: Blockbuster Dynamics in an Ailing Economy."
In its brief overview,
the piece mentions the pitfalls and opportunities evident to anyone familiar
with the technology. These include
what Mr. Harris calls an "innovation glut" of good ideas, novel and
ground-breaking technologies, and inspired and dedicated scientists. On the "downside," Mr. Harris writes,
the "technologies are just as complicated as they are inspired," and as a
result, "they tend to take a long time to go from dreams to drugs or to evolve
through the concept-to-currency cycle."
He quotes a term for a successful clinical trial as taking about nine
years, not including discovery, preclinical testing, or the time it takes for
the Food and Drug Administration to approve a biologic drug after filing of a
BLA.
Despite these
difficulties, the future looks promising to Mr. Harris:
The
truth is, people will continue to be susceptible to disease and drug
development innovation, an aesthetic dynamic, unlike material products, that
will never fall out of favor. Investors know that ideas are the primary origins
of drug development and that foundation must be nurtured with money in order to
cultivate the process to a marketed stage.
The proof is in the
biologic drug pipeline: the
biotech industry has already produced "more than 200 prescription [drug]
products, hundreds of diagnostics tools and tests and also has more than 400
candidates in clinical trials." The
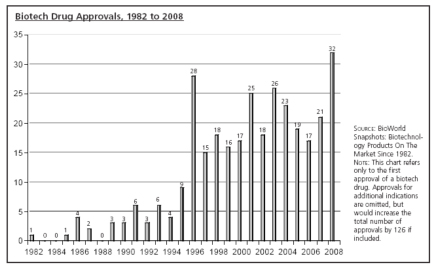
following chart illustrates these successes:
As shown, while the
number of approved biotechnology-based products approved per year is variable,
the trend is upward. Mr. Harris
characterized biotechnology drugs as being the fastest-growing sector for drug
development, and predicts that biotech drugs will comprise over 50% of all drug
approvals by 2015 and more than 75% by 2025. These predictions are supported by the expected benefits of
increased understanding of drug targets and the molecular and genetic bases of
disease, as well as the declining conventional small-molecule drug pipelines in
most major pharmaceutical companies.
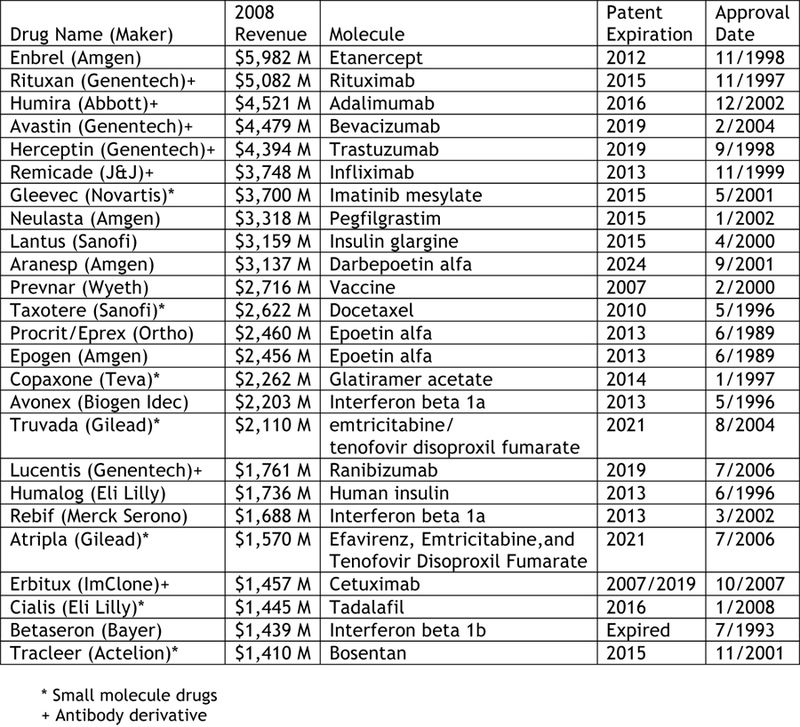
The table
below represents information from Mr. Harris' article, setting forth the revenues
for each of the 25 top biotechnology drugs in 2008, and in addition includes
the date each drug product was first approved by the FDA and when patents
protecting each drug are due to expire. It should be kept in mind that one feature of all these drugs is that
they have been approved for more than one indication; indeed, Mr. Harris reports that Genentech's Avastin is being
tested in more than 450 clinical trials for treating more than 30 different
types of cancer. It should also be
kept in mind that 7 of the 25 "biotech" drugs are small molecules, and another
6 are antibodies. We leave it as
an exercise for the reader to assess the relative levels of protection provided
by patents and data exclusivity should Congress pass a follow-on biologics bill
this year.
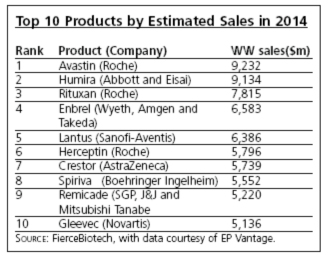
Finally,
the article includes a prediction of the estimated sales and the identities of
the top 10 biotech drugs expected in 2014 (see table below). Eight of the ten are top 25 biotech drugs today, with the
other two being the small molecule drugs Crestor (rosuvastatin, used for lowering
cholesterol) and Spiriva (tiotropium, a
bronchodilator for treating asthma and COPD).
This list is the same as the list compiled by EvaluatePharma, a market forecasting firm
specializing in the life sciences sector, earlier in this year (see
"Future Drug Sales Predictions Highlight
Importance of Follow-on Biologics Legislation"). Interestingly, the top three current
drugs reported by EvaluatePharma (Lipitor, Plavix, and Advair) are all small
molecule drugs having higher revenues than the highest biologic drug, Enbrel
(Lipitor's revenues are more than double Enbrel's), and half the top ten in
that survey are small molecule drugs and three are antibodies; Amgen's Epogen
and Enbrel are the only recombinant drug products in EvaluatePharma's top
ten. In contrast, 11 of the 19
drugs in BioWorld's top 25 are recombinant products.
The BioWorld report follows other recent reports
indicating that the biotechnology sector continues to improve, weathering
serious threats caused by the economic downturn last year (see "Biotech/Pharma Financing Improving, R&D Spending Up"; "Investors Saw Biotech Rebound Coming"; and "Is Biotech/Pharma Beginning to Bounce Back?"). In view of the sentiments expressed by
Mr. Harris about the reality that the need for new drugs is never-ending, that
constitutes good news for biotech and pharma companies and the public combined.

Leave a comment